Brainstem serotonin deficiency in the neonatal period: autonomic dysregulation during mild cold stress
- PMID: 21486799
- PMCID: PMC3090603
- DOI: 10.1113/jphysiol.2010.203679
Brainstem serotonin deficiency in the neonatal period: autonomic dysregulation during mild cold stress
Abstract
Based on previous studies in adult animals, devoid of 5-HT neurones, showing altered thermoregulation in cold stress (4°C) and a reduced ventilatory response to CO₂, we hypothesized that neonatal mice lacking 60-70% of their 5-HT neurones (Pet-1(-/-)) would have: (1) a reduced thermogenic response to a mild drop in ambient temperature (TA), (2) reduced V(E) and heart rate (HR) responses to mild cooling that reflect this reduced thermogenic response, and (3) a reduced ventilatory response to CO₂ after postnatal day 12 (P12), when 5-HT neurones become chemosensitive in vitro. We first determined that a 60-70% loss of 5-HT-positive neurones results in a ~90% loss of 5-HT from the brainstems of Pet-1(-/-) animals. We then subjected Pet-1(-/-) and wild-type (WT) mice (N = 5) to mild environmental cooling (T(A) = 29°C) at ~P12. T(A) was initially held at 34°C for ~20 min, reduced to 29°C over 15 min and held for an additional 10 min at steady state, and then returned to 34°C. From 34°C to 29°C, there was a robust increase in V(O₂) in P12WT, but not Pet-1(-/-) animals (68±19.9% versus -16±8%, respectively; P = 0.002). On average, body temperature (T(B)) dropped 1.1°C more in Pet-1(-/-) compared to WT animals (P = 0.03). HR remained unchanged in WT but dropped 22±2.3% in Pet-1(-/-) animals (P = 0.01). Genotype had no effect on tail temperature (T(T)), either at 34°C or 29°C. After cooling, values for V(O₂) and HR of Pet-1(-/-) animals were no different to values predicted by Q₁₀ effects alone, while values of WT animals were greater than predicted. V(E) increased in WT with cooling, while it decreased in Pet-1(-/-) animals (P = 0.002). Still, Pet-1(-/-) animals hyperventilated relative to WT (increased V(E)/V(O₂)) irrespective of T(A) (P = 0.002). As tested in a separate group of pups, there was no difference in the ventilatory response to CO₂ between WT and Pet-1(-/-) animals, either at P5 or P15. We conclude that during neonatal life in mouse pups: (1) brainstem 5-HT is critical for the thermogenic response to a mild drop in environmental temperature probably via a sympathetically-mediated increase in brown fat metabolism; (2) reduced thermogenesis probably contributes to the reduced HR and V(O₂) observed with 5-HT deficiency; and (3) the presence of some brainstem 5-HT is sufficient for an appropriate ventilatory response to hypercapnia up until P15. Infants with reduced brainstem 5-HT could be prone to cardiovascular and respiratory abnormalities resulting from compromised thermogenesis.
Figures
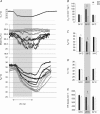
 is not significantly different in Pet-1−/− animals compared to WT. The disparity between genotypes becomes apparent as TA is reduced to 29°C (shaded region). C, at 34°C, TB is slightly but significantly lower in Pet-1−/− animals compared to WT. As is the case with
is not significantly different in Pet-1−/− animals compared to WT. The disparity between genotypes becomes apparent as TA is reduced to 29°C (shaded region). C, at 34°C, TB is slightly but significantly lower in Pet-1−/− animals compared to WT. As is the case with  , the disparity becomes greater with cooling to 29°C. D, there were no differences in tail temperature (TT) between Pet-1−/− and WT animals at either TA. Note: TT is less than 29°C because of a TA gradient within the chamber (TA was 1.5°C lower at the tail end compared to the middle). E, there is a significantly greater reduction in HR with cooling in Pet-1−/− animals compared to WT. All data in B–E are means ± SE. *Significant genotype effect, P < 0.05; †genotype ×TA interaction, P < 0.05.
, the disparity becomes greater with cooling to 29°C. D, there were no differences in tail temperature (TT) between Pet-1−/− and WT animals at either TA. Note: TT is less than 29°C because of a TA gradient within the chamber (TA was 1.5°C lower at the tail end compared to the middle). E, there is a significantly greater reduction in HR with cooling in Pet-1−/− animals compared to WT. All data in B–E are means ± SE. *Significant genotype effect, P < 0.05; †genotype ×TA interaction, P < 0.05.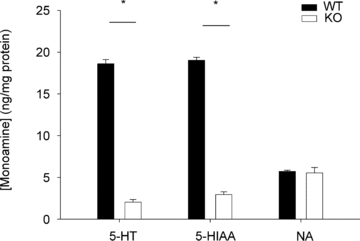
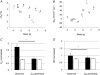
 (2-factor repeated-measures ANOVA: P < 0.05 for each). C and D,
(2-factor repeated-measures ANOVA: P < 0.05 for each). C and D,  and HR after cooling (normalised to baseline values) for WT (filled bars), and Pet-1−/− (open bars). Shown are observed values and values predicted from the Q10 effect alone (assuming Q10 co-efficient of 2 and with a drop in TB of 4.2°C in Pet-1−/− and 3.1°C in WT). Data in C and D are means ± SE. *Significant difference exists between observed WT values for
and HR after cooling (normalised to baseline values) for WT (filled bars), and Pet-1−/− (open bars). Shown are observed values and values predicted from the Q10 effect alone (assuming Q10 co-efficient of 2 and with a drop in TB of 4.2°C in Pet-1−/− and 3.1°C in WT). Data in C and D are means ± SE. *Significant difference exists between observed WT values for  and HR and the values predicted from the Q10 effect alone. No such difference exists between the observed and predicted values for Pet-1−/− animals.
and HR and the values predicted from the Q10 effect alone. No such difference exists between the observed and predicted values for Pet-1−/− animals.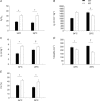
 is elevated in P12 Pet-1−/− animals, irrespective of TA. B, from TA= 34°C to 29°C,
is elevated in P12 Pet-1−/− animals, irrespective of TA. B, from TA= 34°C to 29°C,  decreases in Pet-1−/− animals, but increases in WT animals. C and D,
decreases in Pet-1−/− animals, but increases in WT animals. C and D,  is selectively reduced in Pet-1−/− animals from a reduction in tidal volume (VT) and respiratory frequency (f) E, respiratory variability, as measured by the co-efficient of variation in the respiratory period (CV%) is higher in Pet-1−/− animals at both TA. All data are means ± SE. *Significant genotype effect, P < 0.05; †genotype ×TA interaction, P < 0.05.
is selectively reduced in Pet-1−/− animals from a reduction in tidal volume (VT) and respiratory frequency (f) E, respiratory variability, as measured by the co-efficient of variation in the respiratory period (CV%) is higher in Pet-1−/− animals at both TA. All data are means ± SE. *Significant genotype effect, P < 0.05; †genotype ×TA interaction, P < 0.05.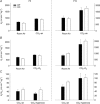
 of WT and Pet-1−/− (KO) animals at P5 and P15, during room air and after 2 min of 5% CO2 balanced with air (CO2–air). B,
of WT and Pet-1−/− (KO) animals at P5 and P15, during room air and after 2 min of 5% CO2 balanced with air (CO2–air). B,  of WT and Pet-1−/− animals during room air and after 2 min of 5% CO2 balanced with O2 (CO2–O2). C, increase in
of WT and Pet-1−/− animals during room air and after 2 min of 5% CO2 balanced with O2 (CO2–O2). C, increase in  (% change) after 2 min of either CO2–air or CO2–O2 in each genotype at P5 and P15. All data are means ± SE.
(% change) after 2 min of either CO2–air or CO2–O2 in each genotype at P5 and P15. All data are means ± SE.References
-
- Allen GV, Cechetto DF. Serotoninergic and nonserotoninergic neurons in the medullary raphe system have axon collateral projections to autonomic and somatic cell groups in the medulla and spinal cord. J Comp Neurol. 1994;350:357–366. - PubMed
-
- Bowker RM, Westlund KN, Coulter JD. Serotonergic projections to the spinal cord from the midbrain in the rat: an immunocytochemical and retrograde transport study. Neurosci Lett. 1981;24:221–226. - PubMed
-
- Brown JW, Sirlin EA, Benoit AM, Hoffman JM, Darnall RA. Activation of 5-HT1A receptors in medullary raphe disrupts sleep and decreases shivering during cooling in the conscious piglet. Am J Physiol Regul Integr Comp Physiol. 2008;294:R884–R894. - PubMed
-
- Clancy B, Darlington RB, Finlay BL. Translating developmental time across mammalian species. Neuroscience. 2001;105:7–17. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

