Scapinin-induced inhibition of axon elongation is attenuated by phosphorylation and translocation to the cytoplasm
- PMID: 21487013
- PMCID: PMC3103351
- DOI: 10.1074/jbc.M110.205781
Scapinin-induced inhibition of axon elongation is attenuated by phosphorylation and translocation to the cytoplasm
Abstract
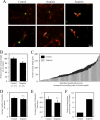
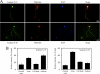
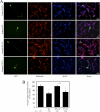
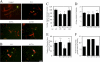
Scapinin is an actin- and PP1-binding protein that is exclusively expressed in the brain; however, its function in neurons has not been investigated. Here we show that expression of scapinin in primary rat cortical neurons inhibits axon elongation without affecting axon branching, dendritic outgrowth, or polarity. This inhibitory effect was dependent on its ability to bind actin because a mutant form that does not bind actin had no effect on axon elongation. Immunofluorescence analysis showed that scapinin is predominantly located in the distal axon shaft, cell body, and nucleus of neurons and displays a reciprocal staining pattern to phalloidin, consistent with previous reports that it binds actin monomers to inhibit polymerization. We show that scapinin is phosphorylated at a highly conserved site in the central region of the protein (Ser-277) by Cdk5 in vitro. Expression of a scapinin phospho-mimetic mutant (S277D) restored normal axon elongation without affecting actin binding. Instead, phosphorylated scapinin was sequestered in the cytoplasm of neurons and away from the axon. Because its expression is highest in relatively plastic regions of the adult brain (cortex, hippocampus), scapinin is a new regulator of neurite outgrowth and neuroplasticity in the brain.
Figures



Similar articles
-
Neuron-enriched phosphatase and actin regulator 3 (Phactr3)/ nuclear scaffold-associated PP1-inhibiting protein (Scapinin) regulates dendritic morphology via its protein phosphatase 1-binding domain.Biochem Biophys Res Commun. 2020 Jul 23;528(2):322-329. doi: 10.1016/j.bbrc.2020.05.006. Epub 2020 May 15. Biochem Biophys Res Commun. 2020. PMID: 32423795
-
Phosphorylation by PKA and Cdk5 Mediates the Early Effects of Synapsin III in Neuronal Morphological Maturation.J Neurosci. 2015 Sep 23;35(38):13148-59. doi: 10.1523/JNEUROSCI.1379-15.2015. J Neurosci. 2015. PMID: 26400944 Free PMC article.
-
c-Jun NH2-terminal kinase (JNK)-interacting protein-3 (JIP3) regulates neuronal axon elongation in a kinesin- and JNK-dependent manner.J Biol Chem. 2013 May 17;288(20):14531-14543. doi: 10.1074/jbc.M113.464453. Epub 2013 Apr 10. J Biol Chem. 2013. PMID: 23576431 Free PMC article.
-
Scapinin, the protein phosphatase 1 binding protein, enhances cell spreading and motility by interacting with the actin cytoskeleton.PLoS One. 2009;4(1):e4247. doi: 10.1371/journal.pone.0004247. Epub 2009 Jan 22. PLoS One. 2009. PMID: 19158953 Free PMC article.
-
Phosphorylation of Drebrin and Its Role in Neuritogenesis.Adv Exp Med Biol. 2017;1006:49-60. doi: 10.1007/978-4-431-56550-5_4. Adv Exp Med Biol. 2017. PMID: 28865014 Review.
Cited by
-
Genome-wide analysis of H4K5 acetylation associated with fear memory in mice.BMC Genomics. 2013 Aug 8;14:539. doi: 10.1186/1471-2164-14-539. BMC Genomics. 2013. PMID: 23927422 Free PMC article.
-
A firm grip does not always pay off: a new Phact(r) 4 integrin signaling.Genes Dev. 2012 Jan 1;26(1):1-5. doi: 10.1101/gad.184192.111. Genes Dev. 2012. PMID: 22215804 Free PMC article.
-
Identifying a kinase network regulating FGF14:Nav1.6 complex assembly using split-luciferase complementation.PLoS One. 2015 Feb 6;10(2):e0117246. doi: 10.1371/journal.pone.0117246. eCollection 2015. PLoS One. 2015. PMID: 25659151 Free PMC article.
-
Case report: Early-onset parkinsonism among the neurological features in children with PHACTR1 variants.Front Neurol. 2023 Jul 6;14:1181015. doi: 10.3389/fneur.2023.1181015. eCollection 2023. Front Neurol. 2023. PMID: 37483454 Free PMC article.
-
PPARgamma agonists rescue increased phosphorylation of FGF14 at S226 in the Tg2576 mouse model of Alzheimer's disease.Exp Neurol. 2017 Sep;295:1-17. doi: 10.1016/j.expneurol.2017.05.005. Epub 2017 May 15. Exp Neurol. 2017. PMID: 28522250 Free PMC article.
References
-
- Sagara J., Higuchi T., Hattori Y., Moriya M., Sarvotham H., Shima H., Shirato H., Kikuchi K., Taniguchi S. (2003) J. Biol. Chem. 278, 45611–45619 - PubMed
-
- Favot L., Gillingwater M., Scott C., Kemp P. R. (2005) FEBS Lett. 579, 100–104 - PubMed
-
- Worch S., Fiedler E., Hansmann I., Schlote D. (2006) Cytogenet. Genome Res. 115, 23–29 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

