Activating PKC-β1 at the blood-brain barrier reverses induction of P-glycoprotein activity by dioxin and restores drug delivery to the CNS
- PMID: 21487415
- PMCID: PMC3130326
- DOI: 10.1038/jcbfm.2011.44
Activating PKC-β1 at the blood-brain barrier reverses induction of P-glycoprotein activity by dioxin and restores drug delivery to the CNS
Abstract
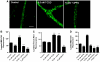
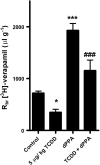
Upregulation of blood-brain barrier (BBB) P-glycoprotein expression causes central nervous system (CNS) pharmacoresistance. However, activation of BBB protein kinase C-β1 (PKC-β1) rapidly reduces basal P-glycoprotein transport activity. We tested whether PKC-β1 activation would reverse CNS drug resistance caused by dioxin acting through aryl hydrocarbon receptor. A selective PKC-β1 agonist abolished the increase in P-glycoprotein activity induced by dioxin in isolated rat brain capillaries and reversed the effect of dioxin on brain uptake of verapamil in dioxin-dosed rats. Thus, targeting BBB PKC-β1 may be an effective strategy to improve drug delivery to the brain, even in drug-resistant individuals.
Figures
References
-
- Bauer B, Hartz AM, Fricker G, Miller DS. Pregnane X receptor up-regulation of P-glycoprotein expression and transport function at the blood-brain barrier. Mol Pharmacol. 2004;66:413–419. - PubMed
-
- Bauer B, Hartz AM, Lucking JR, Yang X, Pollack GM, Miller DS. Coordinated nuclear receptor regulation of the efflux transporter, Mrp2, and the phase-II metabolizing enzyme, GSTpi, at the blood-brain barrier. J Cereb Blood Flow Metab. 2008;28:1222–1234. - PubMed
-
- Hartz AM, Bauer B, Fricker G, Miller DS. Rapid regulation of P-glycoprotein at the blood-brain barrier by endothelin-1. Mol Pharmacol. 2004;66:387–394. - PubMed
-
- Hartz AM, Bauer B, Fricker G, Miller DS. Rapid modulation of P-glycoprotein-mediated transport at the blood-brain barrier by tumor necrosis factor-alpha and lipopolysaccharide. Mol Pharmacol. 2006;69:462–470. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

