Mammalian peroxisomal ABC transporters: from endogenous substrates to pathology and clinical significance
- PMID: 21488864
- PMCID: PMC3246701
- DOI: 10.1111/j.1476-5381.2011.01435.x
Mammalian peroxisomal ABC transporters: from endogenous substrates to pathology and clinical significance
Abstract
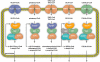
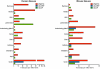
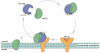
Peroxisomes are indispensable organelles in higher eukaryotes. They are essential for a number of important metabolic pathways, including fatty acid α- and β-oxidation, and biosynthesis of etherphospholipids and bile acids. However, the peroxisomal membrane forms an impermeable barrier to these metabolites. Therefore, peroxisomes need specific transporter proteins to transfer these metabolites across their membranes. The mammalian peroxisomal membrane harbours three ATP-binding cassette (ABC) transporters. In recent years, significant progress has been made in unravelling the functions of these ABC transporters. There is ample evidence that they are involved in the transport of very long-chain fatty acids, pristanic acid, di- and trihydroxycholestanoic acid, dicarboxylic acids and tetracosahexaenoic acid (C24:6ω3). Surprisingly, only one disease is associated with a deficiency of a peroxisomal ABC transporter. Mutations in the ABCD1 gene encoding the peroxisomal ABC transporter adrenoleukodystrophy protein are the cause for X-linked adrenoleukodystrophy, an inherited metabolic storage disorder. This review describes the current state of knowledge on the mammalian peroxisomal ABC transporters with a particular focus on their function in metabolite transport.
© 2011 Academic Medical Center & Rothamsted Research Ltd. British Journal of Pharmacology © 2011 The British Pharmacological Society.
Figures


References
-
- Albet S, Causeret C, Bentejac M, Mandel JL, Aubourg P, Maurice B. Fenofibrate differently alters expression of genes encoding ATP-binding transporter proteins of the peroxisomal membrane. FEBS Lett. 1997;405:394–397. - PubMed
-
- Antonenkov VD, Hiltunen JK. Peroxisomal membrane permeability and solute transfer. Biochim Biophys Acta. 2006;1763:1697–1706. - PubMed
-
- Antonenkov VD, Grunau S, Ohlmeier S, Hiltunen JK. Peroxisomes are oxidative organelles. Antioxid Redox Signal. 2010;13:525–537. - PubMed
-
- Aubourg P, Dubois-Dalcq M. X-linked adrenoleukodystrophy enigma: how does the ALD peroxisomal transporter mutation affect CNS glia? Glia. 2000;29:186–190. - PubMed
-
- Berger J, Albet S, Bentejac M, Netik A, Holzinger A, Roscher AA, et al. The four murine peroxisomal ABC-transporter genes differ in constitutive, inducible and developmental expression. Eur J Biochem. 1999;265:719–727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

