High-resolution secondary ion mass spectrometry reveals the contrasting subcellular distribution of arsenic and silicon in rice roots
- PMID: 21490163
- PMCID: PMC3177285
- DOI: 10.1104/pp.111.173088
High-resolution secondary ion mass spectrometry reveals the contrasting subcellular distribution of arsenic and silicon in rice roots
Abstract
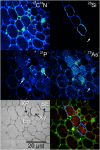
Rice (Oryza sativa) takes up arsenite mainly through the silicic acid transport pathway. Understanding the uptake and sequestration of arsenic (As) into the rice plant is important for developing strategies to reduce As concentration in rice grain. In this study, the cellular and subcellular distributions of As and silicon (Si) in rice roots were investigated using high-pressure freezing, high-resolution secondary ion mass spectrometry, and transmission electron microscopy. Rice plants, both the lsi2 mutant lacking the Si/arsenite efflux transporter Lsi2 and its wild-type cultivar, with or without an iron plaque, were treated with arsenate or arsenite. The formation of iron plaque on the root surface resulted in strong accumulation of As and phosphorous on the epidermis. The lsi2 mutant showed stronger As accumulation in the endodermal vacuoles, where the Lsi2 transporter is located in the plasma membranes, than the wild-type line. As also accumulated in the vacuoles of some xylem parenchyma cells and in some pericycle cells, particularly in the wild-type mature root zone. Vacuolar accumulation of As is associated with sulfur, suggesting that As may be stored as arsenite-phytochelatin complexes. Si was localized in the cell walls of the endodermal cells with little apparent effect of the Lsi2 mutation on its distribution. This study reveals the vacuolar sequestration of As in rice roots and contrasting patterns of As and Si subcellular localization, despite both being transported across the plasma membranes by the same transporters.
Figures







References
-
- Ali M, Badruzzaman A, Jalil M, Hossain M, Ahmed M, Al Masud A, Kamruzzaman M, Azizur Rahman M. (2003) Fate of arsenic extracted with groundwater. Ahmed M, ed, Fate of Arsenic in the Environment. International Training Network, Dhaka, Bangladesh, pp 7–20
-
- Armstrong W. (1967) Oxidising activity of roots in waterlogged soils. Physiol Plant 20: 920–926
-
- Bleeker PM, Hakvoort HWJ, Bliek M, Souer E, Schat H. (2006) Enhanced arsenate reduction by a CDC25-like tyrosine phosphatase explains increased phytochelatin accumulation in arsenate-tolerant Holcus lanatus. Plant J 45: 917–929 - PubMed
-
- Bogdan K, Schenk MK. (2008) Arsenic in rice (Oryza sativa L.) related to dynamics of arsenic and silicic acid in paddy soils. Environ Sci Technol 42: 7885–7890 - PubMed
-
- Brammer H, Ravenscroft P. (2009) Arsenic in groundwater: a threat to sustainable agriculture in South and South-east Asia. Environ Int 35: 647–654 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

