Mixed mode oscillations in mouse spinal motoneurons arise from a low excitability state
- PMID: 21490224
- PMCID: PMC6622841
- DOI: 10.1523/JNEUROSCI.6363-10.2011
Mixed mode oscillations in mouse spinal motoneurons arise from a low excitability state
Abstract
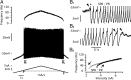
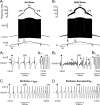
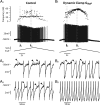
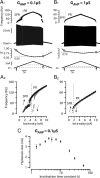
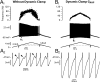
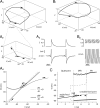
We explain the mechanism that elicits the mixed mode oscillations (MMOs) and the subprimary firing range that we recently discovered in mouse spinal motoneurons. In this firing regime, high-frequency subthreshold oscillations appear a few millivolts below the spike voltage threshold and precede the firing of a full blown spike. By combining intracellular recordings in vivo (including dynamic clamp experiments) in mouse spinal motoneurons and modeling, we show that the subthreshold oscillations are due to the spike currents and that MMOs appear each time the membrane is in a low excitability state. Slow kinetic processes largely contribute to this low excitability. The clockwise hysteresis in the I-F relationship, frequently observed in mouse motoneurons, is mainly due to a substantial slow inactivation of the sodium current. As a consequence, less sodium current is available for spiking. This explains why a large subprimary range with numerous oscillations is present in motoneurons displaying a clockwise hysteresis. In motoneurons whose I-F curve exhibits a counterclockwise hysteresis, it is likely that the slow inactivation operates on a shorter time scale and is substantially reduced by the de-inactivating effect of the afterhyperpolarization (AHP) current, thus resulting in a more excitable state. This accounts for the short subprimary firing range with only a few MMOs seen in these motoneurons. Our study reveals a new role for the AHP current that sets the membrane excitability level by counteracting the slow inactivation of the sodium current and allows or precludes the appearance of MMOs.
Figures


Similar articles
-
The Subprimary Range of Firing Is Present in Both Cat and Mouse Spinal Motoneurons and Its Relationship to Force Development Is Similar for the Two Species.J Neurosci. 2018 Nov 7;38(45):9741-9753. doi: 10.1523/JNEUROSCI.2898-17.2018. Epub 2018 Sep 24. J Neurosci. 2018. PMID: 30249797 Free PMC article.
-
Fast kinetics, high-frequency oscillations, and subprimary firing range in adult mouse spinal motoneurons.J Neurosci. 2009 Sep 9;29(36):11246-56. doi: 10.1523/JNEUROSCI.3260-09.2009. J Neurosci. 2009. PMID: 19741131 Free PMC article.
-
Persistent sodium currents and repetitive firing in motoneurons of the sacrocaudal spinal cord of adult rats.J Neurophysiol. 2006 Sep;96(3):1141-57. doi: 10.1152/jn.00335.2005. Epub 2005 Nov 9. J Neurophysiol. 2006. PMID: 16282206 Free PMC article.
-
Essential role of the persistent sodium current in spike initiation during slowly rising inputs in mouse spinal neurones.J Physiol. 2006 Aug 1;574(Pt 3):819-34. doi: 10.1113/jphysiol.2006.107094. Epub 2006 May 25. J Physiol. 2006. PMID: 16728453 Free PMC article.
-
Historical reflections on the afterhyperpolarization--firing rate relation of vertebrate spinal neurons.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2007 Feb;193(2):145-58. doi: 10.1007/s00359-006-0198-2. Epub 2006 Dec 28. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2007. PMID: 17192827 Review.
Cited by
-
The Subprimary Range of Firing Is Present in Both Cat and Mouse Spinal Motoneurons and Its Relationship to Force Development Is Similar for the Two Species.J Neurosci. 2018 Nov 7;38(45):9741-9753. doi: 10.1523/JNEUROSCI.2898-17.2018. Epub 2018 Sep 24. J Neurosci. 2018. PMID: 30249797 Free PMC article.
-
The dendritic location of the L-type current and its deactivation by the somatic AHP current both contribute to firing bistability in motoneurons.Front Comput Neurosci. 2014 Jan 27;8:4. doi: 10.3389/fncom.2014.00004. eCollection 2014. Front Comput Neurosci. 2014. PMID: 24478687 Free PMC article.
-
Stochastic and deterministic dynamics of intrinsically irregular firing in cortical inhibitory interneurons.Elife. 2016 Aug 18;5:e16475. doi: 10.7554/eLife.16475. Elife. 2016. PMID: 27536875 Free PMC article.
-
Adult spinal motoneurones are not hyperexcitable in a mouse model of inherited amyotrophic lateral sclerosis.J Physiol. 2014 Apr 1;592(7):1687-703. doi: 10.1113/jphysiol.2013.265843. Epub 2014 Jan 20. J Physiol. 2014. PMID: 24445319 Free PMC article.
-
Effects of persistent inward currents, accommodation, and adaptation on motor unit behavior: a simulation study.J Neurophysiol. 2011 Sep;106(3):1467-79. doi: 10.1152/jn.00419.2011. Epub 2011 Jun 22. J Neurophysiol. 2011. PMID: 21697447 Free PMC article.
References
-
- Abbot LF, Kepler T. Model neurons: from Hodgkin-Huxley to Hopfield. In: Garrido L, editor. Statistical mechanics of neural networks. Berlin: Springer; 1990. pp. 5–18.
-
- Alonso A, Klink R. Differential electroresponsiveness of stellate and pyramidal-like cells of medial entorhinal cortex layer II. J Neurophysiol. 1993;70:128–143. - PubMed
-
- Bennett DJ, Li Y, Siu M. Plateau potentials in sacrocaudal motoneurons of chronic spinal rats, recorded in vitro. J Neurophysiol. 2001;86:1955–1971. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
