Liposomal Tumor Targeting in Drug Delivery Utilizing MMP-2- and MMP-9-Binding Ligands
- PMID: 21490745
- PMCID: PMC3066593
- DOI: 10.1155/2011/160515
Liposomal Tumor Targeting in Drug Delivery Utilizing MMP-2- and MMP-9-Binding Ligands
Abstract
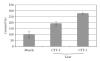
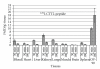
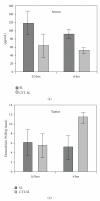
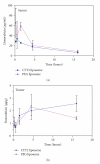
Nanotechnology offers an alternative to conventional treatment options by enabling different drug delivery and controlled-release delivery strategies. Liposomes being especially biodegradable and in most cases essentially nontoxic offer a versatile platform for several different delivery approaches that can potentially enhance the delivery and targeting of therapies to tumors. Liposomes penetrate tumors spontaneously as a result of fenestrated blood vessels within tumors, leading to known enhanced permeability and subsequent drug retention effects. In addition, liposomes can be used to carry radioactive moieties, such as radiotracers, which can be bound at multiple locations within liposomes, making them attractive carriers for molecular imaging applications. Phage display is a technique that can deliver various high-affinity and selectivity peptides to different targets. In this study, gelatinase-binding peptides, found by phage display, were attached to liposomes by covalent peptide-PEG-PE anchor creating a targeted drug delivery vehicle. Gelatinases as extracellular targets for tumor targeting offer a viable alternative for tumor targeting. Our findings show that targeted drug delivery is more efficient than non-targeted drug delivery.
Figures
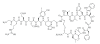

Similar articles
-
Targeted liposomal drug delivery in cancer.Curr Pharm Des. 2004;10(24):2981-9. doi: 10.2174/1381612043383467. Curr Pharm Des. 2004. PMID: 15379663 Review.
-
A liposomal drug platform overrides peptide ligand targeting to a cancer biomarker, irrespective of ligand affinity or density.PLoS One. 2013 Aug 23;8(8):e72938. doi: 10.1371/journal.pone.0072938. eCollection 2013. PLoS One. 2013. PMID: 24009717 Free PMC article.
-
The use of tail-anchored protein chimeras to enhance liposomal cargo delivery.PLoS One. 2019 Feb 22;14(2):e0212701. doi: 10.1371/journal.pone.0212701. eCollection 2019. PLoS One. 2019. PMID: 30794671 Free PMC article.
-
From phage display to nanoparticle delivery: functionalizing liposomes with multivalent peptides improves targeting to a cancer biomarker.Bioconjug Chem. 2013 Jan 16;24(1):85-96. doi: 10.1021/bc300498d. Epub 2013 Jan 7. Bioconjug Chem. 2013. PMID: 23186007 Free PMC article.
-
Pros and cons of the liposome platform in cancer drug targeting.J Liposome Res. 2006;16(3):175-83. doi: 10.1080/08982100600848769. J Liposome Res. 2006. PMID: 16952872 Review.
Cited by
-
Acid-Sphingomyelinase Triggered Fluorescently Labeled Sphingomyelin Containing Liposomes in Tumor Diagnosis after Radiation-Induced Stress.Int J Mol Sci. 2021 Apr 8;22(8):3864. doi: 10.3390/ijms22083864. Int J Mol Sci. 2021. PMID: 33917976 Free PMC article.
-
Mannosylated liposomes for targeted gene delivery.Int J Nanomedicine. 2012;7:1079-89. doi: 10.2147/IJN.S29183. Epub 2012 Feb 22. Int J Nanomedicine. 2012. PMID: 22393297 Free PMC article.
-
Tumor-targeting peptides from combinatorial libraries.Adv Drug Deliv Rev. 2017 Feb;110-111:13-37. doi: 10.1016/j.addr.2016.05.009. Epub 2016 May 19. Adv Drug Deliv Rev. 2017. PMID: 27210583 Free PMC article. Review.
-
Insights on the Dynamic Innovative Tumor Targeted-Nanoparticles-Based Drug Delivery Systems Activation Techniques.Int J Nanomedicine. 2022 Dec 7;17:6131-6155. doi: 10.2147/IJN.S386037. eCollection 2022. Int J Nanomedicine. 2022. PMID: 36514378 Free PMC article. Review.
-
In vivo targeting of activated leukocytes by a β2-integrin binding peptide.Mol Diagn Ther. 2014 Feb;18(1):39-44. doi: 10.1007/s40291-013-0052-5. Mol Diagn Ther. 2014. PMID: 23982749
References
-
- Torchilin V, Weissig V. Liposomes: A Practical Approach. 2nd edition. Oxford, UK: Oxford University Press; 2003.
-
- Bendas G. Immunoliposomes: a promising approach to targeting cancer therapy. BioDrugs. 2001;15(4):215–224. - PubMed
-
- Nagaset H, Woessner JF., Jr. Matrix metalloproteinases. Minireview. The Journal of Biological Chemistry. 1999;274(31):21491–21494. - PubMed
-
- Curran S, Murray GI. Matrix metalloproteinasesmolecular aspects of their roles in tumour invasion and metastasis. European Journal of Cancer. 2000;36(13):1621–1630. - PubMed
-
- McCawley LJ, Matrisian LM. Matrix metalloproteinases: they’re not just for matrix anymore! Current Opinion in Cell Biology. 2001;13(5):534–540. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

