The exocyst protein Sec10 interacts with Polycystin-2 and knockdown causes PKD-phenotypes
- PMID: 21490950
- PMCID: PMC3072367
- DOI: 10.1371/journal.pgen.1001361
The exocyst protein Sec10 interacts with Polycystin-2 and knockdown causes PKD-phenotypes
Abstract
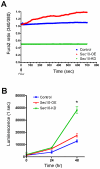
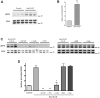
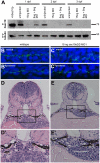
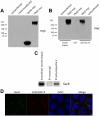
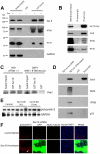
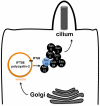
Autosomal dominant polycystic kidney disease (ADPKD) is characterized by formation of renal cysts that destroy the kidney. Mutations in PKD1 and PKD2, encoding polycystins-1 and -2, cause ADPKD. Polycystins are thought to function in primary cilia, but it is not well understood how these and other proteins are targeted to cilia. Here, we provide the first genetic and biochemical link between polycystins and the exocyst, a highly-conserved eight-protein membrane trafficking complex. We show that knockdown of exocyst component Sec10 yields cellular phenotypes associated with ADPKD, including loss of flow-generated calcium increases, hyperproliferation, and abnormal activation of MAPK. Sec10 knockdown in zebrafish phenocopies many aspects of polycystin-2 knockdown-including curly tail up, left-right patterning defects, glomerular expansion, and MAPK activation-suggesting that the exocyst is required for pkd2 function in vivo. We observe a synergistic genetic interaction between zebrafish sec10 and pkd2 for many of these cilia-related phenotypes. Importantly, we demonstrate a biochemical interaction between Sec10 and the ciliary proteins polycystin-2, IFT88, and IFT20 and co-localization of the exocyst and polycystin-2 at the primary cilium. Our work supports a model in which the exocyst is required for the ciliary localization of polycystin-2, thus allowing for polycystin-2 function in cellular processes.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Cdc42 deficiency causes ciliary abnormalities and cystic kidneys.J Am Soc Nephrol. 2013 Sep;24(9):1435-50. doi: 10.1681/ASN.2012121236. Epub 2013 Jun 13. J Am Soc Nephrol. 2013. PMID: 23766535 Free PMC article.
-
The small GTPase Cdc42 is necessary for primary ciliogenesis in renal tubular epithelial cells.J Biol Chem. 2011 Jun 24;286(25):22469-77. doi: 10.1074/jbc.M111.238469. Epub 2011 May 4. J Biol Chem. 2011. PMID: 21543338 Free PMC article.
-
The exocyst protein Sec10 is necessary for primary ciliogenesis and cystogenesis in vitro.Mol Biol Cell. 2009 May;20(10):2522-9. doi: 10.1091/mbc.e08-07-0772. Epub 2009 Mar 18. Mol Biol Cell. 2009. PMID: 19297529 Free PMC article.
-
Regulation of polycystin expression, maturation and trafficking.Cell Signal. 2020 Aug;72:109630. doi: 10.1016/j.cellsig.2020.109630. Epub 2020 Apr 8. Cell Signal. 2020. PMID: 32275942 Free PMC article. Review.
-
Cilia and polycystic kidney disease.Semin Cell Dev Biol. 2021 Feb;110:139-148. doi: 10.1016/j.semcdb.2020.05.003. Epub 2020 May 28. Semin Cell Dev Biol. 2021. PMID: 32475690 Review.
Cited by
-
Regulation of Cell Polarity by Exocyst-Mediated Trafficking.Cold Spring Harb Perspect Biol. 2018 Mar 1;10(3):a031401. doi: 10.1101/cshperspect.a031401. Cold Spring Harb Perspect Biol. 2018. PMID: 28264817 Free PMC article. Review.
-
New frontiers: discovering cilia-independent functions of cilia proteins.EMBO Rep. 2015 Oct;16(10):1275-87. doi: 10.15252/embr.201540632. Epub 2015 Sep 9. EMBO Rep. 2015. PMID: 26358956 Free PMC article. Review.
-
A genetic screen in Drosophila reveals an unexpected role for the KIP1 ubiquitination-promoting complex in male fertility.PLoS Genet. 2020 Dec 30;16(12):e1009217. doi: 10.1371/journal.pgen.1009217. eCollection 2020 Dec. PLoS Genet. 2020. PMID: 33378371 Free PMC article.
-
Loss-of-function of the ciliopathy protein Cc2d2a disorganizes the vesicle fusion machinery at the periciliary membrane and indirectly affects Rab8-trafficking in zebrafish photoreceptors.PLoS Genet. 2017 Dec 27;13(12):e1007150. doi: 10.1371/journal.pgen.1007150. eCollection 2017 Dec. PLoS Genet. 2017. PMID: 29281629 Free PMC article.
-
Early insights into the role of Exoc6B associated with spondyloepimetaphyseal dysplasia with joint laxity type 3 in primary ciliogenesis and chondrogenic differentiation in vitro.Mol Biol Rep. 2024 Feb 2;51(1):274. doi: 10.1007/s11033-023-09114-9. Mol Biol Rep. 2024. PMID: 38305850
References
-
- Smyth BJ, Snyder R, Balkovetz DF, Lipschutz JH. Recent advances in the cell biology of polycystic kidney disease. In: Jeon KW, editor. Int Rev Cytol. San Diego: Elsevier Inc; 2003. pp. 52–89. - PubMed
-
- Consortium TIPD. Polycystic kidney disease: The complete structure of the PKD1 gene and its protein. Cell. 1995;81:289–298. - PubMed
-
- Mochizuki T, Wu G, Hayashi T, Xenophontos SL, Veldhuisen B, et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science. 1996;272:1339–1342. - PubMed
-
- Vassilev PM, Guo L, Chen XZ, Segal Y, Peng JB, et al. Polycystin-2 is a novel cation channel implicated in defective intracellular Ca(2+) homeostasis in polycystic kidney disease. Biochem Biophys Res Commun. 2001;282:341–350. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

