SUMO-interacting motifs of human TRIM5α are important for antiviral activity
- PMID: 21490953
- PMCID: PMC3072370
- DOI: 10.1371/journal.ppat.1002019
SUMO-interacting motifs of human TRIM5α are important for antiviral activity
Abstract
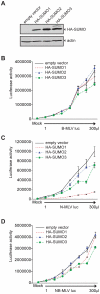
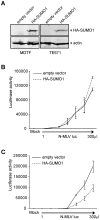
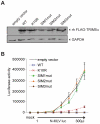
Human TRIM5α potently restricts particular strains of murine leukemia viruses (the so-called N-tropic strains) but not others (the B- or NB-tropic strains) during early stages of infection. We show that overexpression of SUMO-1 in human 293T cells, but not in mouse MDTF cells, profoundly blocks N-MLV infection. This block is dependent on the tropism of the incoming virus, as neither B-, NB-, nor the mutant R110E of N-MLV CA (a B-tropic switch) are affected by SUMO-1 overexpression. The block occurred prior to reverse transcription and could be abrogated by large amounts of restricted virus. Knockdown of TRIM5α in 293T SUMO-1-overexpressing cells resulted in ablation of the SUMO-1 antiviral effects, and this loss of restriction could be restored by expression of a human TRIM5α shRNA-resistant plasmid. Amino acid sequence analysis of human TRIM5α revealed a consensus SUMO conjugation site at the N-terminus and three putative SUMO interacting motifs (SIMs) in the B30.2 domain. Mutations of the TRIM5α consensus SUMO conjugation site did not affect the antiviral activity of TRIM5α in any of the cell types tested. Mutation of the SIM consensus sequences, however, abolished TRIM5α antiviral activity against N-MLV. Mutation of lysines at a potential site of SUMOylation in the CA region of the Gag gene reduced the SUMO-1 block and the TRIM5α restriction of N-MLV. Our data suggest a novel aspect of TRIM5α-mediated restriction, in which the presence of intact SIMs in TRIM5α, and also the SUMO conjugation of CA, are required for restriction. We propose that at least a portion of the antiviral activity of TRIM5α is mediated through the binding of its SIMs to SUMO-conjugated CA.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

