Induction of Plasmodium falciparum-specific CD4+ T cells and memory B cells in Gabonese children vaccinated with RTS,S/AS01(E) and RTS,S/AS02(D)
- PMID: 21494604
- PMCID: PMC3073948
- DOI: 10.1371/journal.pone.0018559
Induction of Plasmodium falciparum-specific CD4+ T cells and memory B cells in Gabonese children vaccinated with RTS,S/AS01(E) and RTS,S/AS02(D)
Abstract
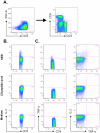
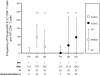
The recombinant circumsporozoite protein (CS) based vaccine, RTS,S, confers protection against Plasmodium falciparum infection in controlled challenge trials and in field studies. The RTS,S recombinant antigen has been formulated with two adjuvant systems, AS01 and AS02, which have both been shown to induce strong specific antibody responses and CD4 T cell responses in adults. As infants and young children are particularly susceptible to malaria infection and constitute the main target population for a malaria vaccine, we have evaluated the induction of adaptive immune responses in young children living in malaria endemic regions following vaccination with RTS,S/AS01(E) and RTS,S/AS02(D). Our data show that a CS-specific memory B cell response is induced one month after the second and third vaccine dose and that CS-specific antibodies and memory B cells persist up to 12 months after the last vaccine injection. Both formulations also induced low but significant amounts of CS-specific IL-2(+) CD4(+) T cells one month after the second and third vaccine dose, upon short-term in vitro stimulation of whole blood cells with peptides covering the entire CS derived sequence in RTS,S. These results provide evidence that both RTS,S/AS01(E) and RTS,S/AS02(D) induced adaptive immune responses including antibodies, circulating memory B cells and CD4(+) T cells directed against P. falciparum CS protein.
Trial registration: ClinicalTrials.gov NCT00307021.
Conflict of interest statement
Figures


References
-
- Garçon N, Heppner DG, Cohen J. Development of RTS,S/AS02: a purified subunit-based malaria vaccine candidate formulated with a novel adjuvant. Expert Rev Vacc. 2003;2:231–238. - PubMed
-
- Gordon DM, McGovern TW, Krzych U, Cohen JC, Schneider I, et al. Safety, immunogenicity, and efficacy of a recombinantly produced Plasmodium falciparum circumsporozoite protein-hepatitis B surface antigen subunit vaccine. J Infect Dis. 1995;171:1576–1585. - PubMed
-
- Kester KE, Cummings JF, Ofori-Anyinam O, Ockenhouse CF, Krzych U, et al. RTS,S Vaccine Evaluation Group. Randomized, double-blind, phase 2a trial of falciparum malaria vaccines RTS,S/AS01B and RTS,S/AS02A in malaria-naive adults: safety, efficacy, and immunologic associates of protection. J Infect Dis. 2009;200:337–346. - PubMed
-
- Stoute JA, Slaoui M, Heppner DG, Momin P, Kester KE, et al. A preliminary evaluation of a recombinant circumsporozoite protein vaccine against Plasmodium falciparum malaria. RTS,S Malaria Vaccine Evaluation Group. N Engl J Med. 1997;336:86–91. - PubMed
-
- Stoute JA, Kester KE, Krzych U, Wellde BT, Hall T, et al. Long term efficacy and immune responses following immunization with the RTS,S malaria vaccine. J Infect Dis. 1998;178:139–1144. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

