Epigenetic regulation of cathepsin L expression in chronic myeloid leukaemia
- PMID: 21496199
- PMCID: PMC4394228
- DOI: 10.1111/j.1582-4934.2010.01203.x
Epigenetic regulation of cathepsin L expression in chronic myeloid leukaemia
Abstract
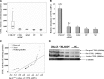
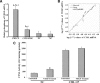
The expression and significance of cathepsin L (CTSL) has been extensively studied in solid tumours. However no such information in chronic myeloid leukaemia (CML) was available. We investigated the activity and expression of this protease in peripheral blood mononuclear cells (PBMCs) of 47 adult CML patients. Thirty adults suffering from systemic diseases and 50 healthy volunteers served as controls. The mRNA levels of CTSL, its specific endogenous inhibitor cystatin C and transcriptional up-regulator vascular endothelial growth factor (VEGF) were quantitated by real-time qPCR. CTSL protease activity and its mRNA expression were significantly higher in CML chronic phase (CP) patients compared to CML accelerated phase/blast crisis (AP/BC) patients and controls (P≤ 0.001). VEGF whose expression was most pronounced in CP and declined (P≤ 0.001) in the advanced phases of the malignancy exhibited a strong positive correlation with CTSL expression (r= 0.97; P≤ 0.001). Cystatin C expression was significantly lower (P≤ 0.001) in CML and displayed inverse correlation with CTSL (r=-0.713; P≤ 0.001) activity. CTSL promoter was significantly hypomethylated in CML CP compared to CML AP/BC patients as well as controls. K562, a BC CML cell line displayed CTSL activity, expression and methylation status of CTSL promoter that was comparable to CML AP/BC patients. Treatment of these cells or PBMCs isolated from CML AP/BC patients with 5'-aza-cytidine resulted in a dramatic increase in CSTL activity and/or expression thereby demonstrating the role of promoter methylation in the stage specific expression of CTSL in CML. Differential expression of CTSL in CML at various stages of malignancy may prove useful in identification of the high-risk patients thereby facilitating better management of disease.
© 2011 The Authors Journal of Cellular and Molecular Medicine © 2011 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures




Similar articles
-
Cathepsins B and L in peripheral blood mononuclear cells of pediatric acute myeloid leukemia: potential poor prognostic markers.Ann Hematol. 2010 Dec;89(12):1223-32. doi: 10.1007/s00277-010-1012-3. Epub 2010 Jun 22. Ann Hematol. 2010. PMID: 20567828
-
[Down-regulation of transcription factor PU.1 via abnormal epigenetic modification in chronic myeloid leukemia].Zhonghua Zhong Liu Za Zhi. 2012 Mar;34(3):169-75. doi: 10.3760/cma.j.issn.0253-3766.2012.03.003. Zhonghua Zhong Liu Za Zhi. 2012. PMID: 22780968 Chinese.
-
Hypomethylation-mediated H19 overexpression increases the risk of disease evolution through the association with BCR-ABL transcript in chronic myeloid leukemia.J Cell Physiol. 2018 Mar;233(3):2444-2450. doi: 10.1002/jcp.26119. Epub 2017 Aug 30. J Cell Physiol. 2018. PMID: 28776669
-
Correlation between preferentially expressed antigen of melanoma and tumour necrosis factor-related apoptosis-inducing ligand gene expression in different types of leukaemia patients.Acta Haematol. 2013;130(4):297-304. doi: 10.1159/000351166. Epub 2013 Aug 31. Acta Haematol. 2013. PMID: 24008770
-
The proto-oncogene expression varies over the course of chronic myeloid leukemia.Hematology. 2008 Feb;13(1):34-40. doi: 10.1179/102453308X315807. Hematology. 2008. PMID: 18534064
Cited by
-
Stress-resistant Translation of Cathepsin L mRNA in Breast Cancer Progression.J Biol Chem. 2015 Jun 19;290(25):15758-15769. doi: 10.1074/jbc.M114.624353. Epub 2015 May 8. J Biol Chem. 2015. PMID: 25957406 Free PMC article.
-
Cathepsin L targeting in cancer treatment.Pharmacol Ther. 2015 Nov;155:105-16. doi: 10.1016/j.pharmthera.2015.08.007. Epub 2015 Aug 20. Pharmacol Ther. 2015. PMID: 26299995 Free PMC article. Review.
-
Cathepsin L in tumor angiogenesis and its therapeutic intervention by the small molecule inhibitor KGP94.Clin Exp Metastasis. 2016 Jun;33(5):461-73. doi: 10.1007/s10585-016-9790-1. Epub 2016 Apr 7. Clin Exp Metastasis. 2016. PMID: 27055649 Free PMC article.
-
In Vitro Study of Cytotoxic Mechanisms of Alkylphospholipids and Alkyltriazoles in Acute Lymphoblastic Leukemia Models.Molecules. 2022 Dec 6;27(23):8633. doi: 10.3390/molecules27238633. Molecules. 2022. PMID: 36500726 Free PMC article.
-
Methyltransferase-like 3 promotes cervical cancer metastasis by enhancing cathepsin L mRNA stability in an N6-methyladenosine-dependent manner.Cancer Sci. 2023 Mar;114(3):837-854. doi: 10.1111/cas.15658. Epub 2023 Jan 2. Cancer Sci. 2023. PMID: 36382580 Free PMC article.
References
-
- Faderl S, Talpaz M, Estrov Z, et al. Chronic myelogenous leukemia: biology and therapy. Ann Intern Med. 1999;131:207–19. - PubMed
-
- Sawyers CL. Chronic myeloid leukemia. N Engl J Med. 1999;340:1330–40. - PubMed
-
- Daley GQ, Van Etten RA, Baltimore D. Induction of chronic myelogenous leukemia in mice by the P210bcr/abl gene of the Philadelphia chromosome. Science. 1990;247:824–30. - PubMed
-
- Heisterkamp N, Jenster G, ten Hoeve J, et al. Acute leukaemia in bcr/abl transgenic mice. Nature. 1990;344:251–3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

