Nrf2 inhibits LXRα-dependent hepatic lipogenesis by competing with FXR for acetylase binding
- PMID: 21504366
- PMCID: PMC6468953
- DOI: 10.1089/ars.2010.3834
Nrf2 inhibits LXRα-dependent hepatic lipogenesis by competing with FXR for acetylase binding
Abstract
Aims: The nuclear receptor liver X receptor-α (LXRα) stimulates lipogenesis, leading to steatosis. Nuclear factor erythroid-2-related factor-2 (Nrf2) contributes to cellular defense mechanism by upregulating antioxidant genes, and may protect the liver from injury inflicted by fat accumulation. However, whether Nrf2 affects LXRα activity is unknown. This study investigated the inhibitory role of Nrf2 in hepatic LXRα activity and the molecular basis.
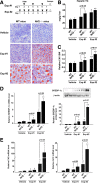
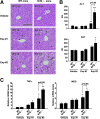
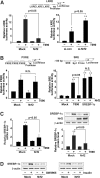
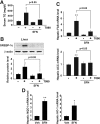
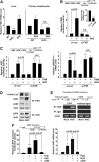
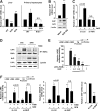
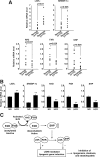
Results: A deficiency of Nrf2 enhanced the ability of LXRα agonist to promote hepatic steatosis, as mediated by lipogenic gene induction. In hepatocytes, Nrf2 overexpression repressed gene transactivation by LXR-binding site activation. Consistently, treatment of mice with sulforaphane (an Nrf2 activator) suppressed T0901317-induced lipogenesis, as confirmed by the experiments using hepatocytes. Nrf2 activation promoted deacetylation of farnesoid X receptor (FXR) by competing for p300, leading to FXR-dependent induction of small heterodimer partner (SHP), which was responsible for the repression of LXRα-dependent gene transcription. In human steatotic samples, the transcript levels of LXRα and SREBP-1 inversely correlated with those of Nrf2, FXR, and SHP.
Innovation: Our findings offer the mechanism to explain how decrease in Nrf2 activity in hepatic steatosis could contribute to the progression of NAFLD, providing the use of Nrf2 as a molecular biomarker to diagnose NAFLD. As certain antioxidants have the abilities to activate Nrf2, clinicians might utilize the activators of Nrf2 as a new therapeutic approach to prevent and/or treat NAFLD.
Conclusion: Nrf2 activation inhibits LXRα activity and LXRα-dependent liver steatosis by competing with FXR for p300, causing FXR activation and FXR-mediated SHP induction. Our findings provide important information on a strategy to prevent and/or treat steatosis.
Figures

References
-
- Brendel C. Schoonjans K. Botrugno OA. Treuter E. Auwerx J. The small heterodimer partner interacts with the liver X receptor alpha and represses its transcriptional activity. Mol Endocrinol. 2002;16:2065–2076. - PubMed
-
- Carmiel-Haggai M. Cederbaum AI. Nieto N. A high-fat diet leads to the progression of non-alcoholic fatty liver disease in obese rats. FASEB J. 2005;19:136–138. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

