Economic considerations of macular edema therapies
- PMID: 21507488
- PMCID: PMC3483086
- DOI: 10.1016/j.ophtha.2010.12.034
Economic considerations of macular edema therapies
Abstract
Purpose: To relate costs and treatment benefits for diabetic macular edema (DME), branch retinal vein occlusion (BRVO), and central retinal vein occlusion (CRVO).
Design: A model of resource use, outcomes, and cost-effectiveness and utility.
Participants: None.
Methods: Results from published clinical trials (index studies) of laser, intravitreal corticosteroids, intravitreal anti-vascular endothelial growth factor (VEGF) agents, and vitrectomy trials were used to ascertain visual benefit and clinical protocols. Calculations followed from the costs of 1 year of treatment for each treatment modality and the visual benefits as ascertained.
Main outcome measures: Visual acuity (VA) saved, cost of therapy, cost per line saved, cost per line-year saved, and costs per quality-adjusted life years (QALYs).
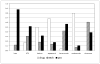
Results: The lines saved for DME (0.26-2.02), BRVO (0.74-4.92), and CRVO (1.2-3.75) yielded calculations of costs/line of saved VA for DME ($1329-$11,609), BRVO ($494-$13,039), and CRVO ($704-$7611); costs/line-year for DME ($60-$561), BRVO ($25-$754), and CRVO ($45-$473); and costs/QALY ($824 to $25,566).
Conclusions: Relative costs and benefits should be considered in perspective when applying and developing treatment strategies.
Copyright © 2011 American Academy of Ophthalmology. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The author does not have any financial interests to disclose.
Figures
Similar articles
-
Clinical applications of cost analysis of diabetic macular edema treatments.Ophthalmology. 2012 Dec;119(12):2558-62. doi: 10.1016/j.ophtha.2012.09.015. Epub 2012 Oct 9. Ophthalmology. 2012. PMID: 23062655
-
Cost-Utility of Anti-Vascular Endothelial Growth Factor Treatment for Macular Edema Secondary to Central Retinal Vein Occlusion.Ophthalmol Retina. 2021 Jul;5(7):656-663. doi: 10.1016/j.oret.2020.09.017. Epub 2020 Sep 28. Ophthalmol Retina. 2021. PMID: 33002672 Free PMC article. Clinical Trial.
-
Comparison of intravitreal bevacizumab upload followed by a dexamethasone implant versus dexamethasone implant monotherapy for retinal vein occlusion with macular edema.Ophthalmologica. 2012;228(2):110-6. doi: 10.1159/000338732. Epub 2012 Jun 23. Ophthalmologica. 2012. PMID: 22739239
-
Steroids and macular edema from retinal vein occlusion.Eur J Ophthalmol. 2011;21 Suppl 6:S37-44. doi: 10.5301/EJO.2010.6053. Eur J Ophthalmol. 2011. PMID: 23264327 Review.
-
Comparison between Ozurdex and intravitreal anti-vascular endothelial growth factor treatment for retinal vein occlusion-related macular edema: A systematic review and meta-analysis of randomized controlled trials.Indian J Ophthalmol. 2019 Nov;67(11):1800-1809. doi: 10.4103/ijo.IJO_382_19. Indian J Ophthalmol. 2019. PMID: 31638037 Free PMC article.
Cited by
-
Corticosteroid use for diabetic macular edema: old fad or new trend?Curr Diab Rep. 2012 Aug;12(4):364-75. doi: 10.1007/s11892-012-0281-8. Curr Diab Rep. 2012. PMID: 22581206 Review.
-
Intravitreal ziv-aflibercept in diabetic vitreous hemorrhage.Int J Retina Vitreous. 2020 Jan 14;6:2. doi: 10.1186/s40942-019-0204-9. eCollection 2020. Int J Retina Vitreous. 2020. PMID: 31956432 Free PMC article. Review.
-
Retinal Vein Occlusion-Background Knowledge and Foreground Knowledge Prospects-A Review.J Clin Med. 2024 Jul 5;13(13):3950. doi: 10.3390/jcm13133950. J Clin Med. 2024. PMID: 38999513 Free PMC article. Review.
-
Collateral Vessel Development in Central and Branch Retinal Vein Occlusions Are Associated With Worse Visual and Anatomic Outcomes.Invest Ophthalmol Vis Sci. 2021 Nov 1;62(14):1. doi: 10.1167/iovs.62.14.1. Invest Ophthalmol Vis Sci. 2021. PMID: 34724540 Free PMC article.
-
Cost-effectiveness of retinal detachment repair.Ophthalmology. 2014 Apr;121(4):946-51. doi: 10.1016/j.ophtha.2013.11.003. Epub 2014 Jan 9. Ophthalmology. 2014. PMID: 24411577 Free PMC article.
References
-
- Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema: Early Treatment Diabetic Retinopathy Study report number 1. Arch Ophthalmol. 1985;103:1796–806. - PubMed
-
- Branch Vein Occlusion Study Group. Argon laser photocoagulation for macular edema in branch retinal vein occlusion. Am J Ophthalmol. 1984;98:271–82. - PubMed
-
- Evaluation of grid pattern photocoagulation for macular edema in central vein occlusion: the Central Vein Occlusion Study Group M report. Ophthalmology. 1995;102:1425–33. - PubMed
-
- Lin JM, Chiu YT, Hung PR, Tsai YY. Early treatment of severe cystoid macular edema in central retinal vein occlusion with posterior sub-Tenon triamcinolone acetonide. Retina. 2007;27:180–9. - PubMed
-
- Nakano S, Yamamoto T, Kirii E, et al. Steroid eye drop treatment (difluprednate ophthalmic emulsion) is effective in reducing refractory diabetic macular edema. Graefes Arch Clin Exp Ophthalmol. 2010;248:805–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

