NF-kappaB-mediated modulation of inducible nitric oxide synthase activity controls induction of the Epstein-Barr virus productive cycle by transforming growth factor beta 1
- PMID: 21507981
- PMCID: PMC3126544
- DOI: 10.1128/JVI.02560-10
NF-kappaB-mediated modulation of inducible nitric oxide synthase activity controls induction of the Epstein-Barr virus productive cycle by transforming growth factor beta 1
Abstract
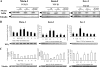
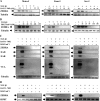
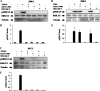
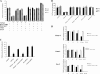
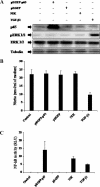
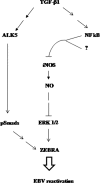
Transforming growth factor beta 1 (TGF-β1) signal transduction has been implicated in many second-messenger pathways, including the NF-κB pathway. We provide evidence of a novel TGF-β1-mediated pathway that leads to extracellular signal-regulated kinase (ERK) 1/2 phosphorylation, which in turn induces expression of an Epstein-Barr virus (EBV) protein, ZEBRA, that is responsible for the induction of the viral lytic cycle. This pathway includes two unexpected steps, both of which are required to control ERK 1/2 phosphorylation: first, a quick and transient activation of NF-κB, and second, downregulation of inducible nitric oxide synthase (iNOS) activity that requires the participation of NF-κB activity. Although necessary, NF-κB alone is not sufficient to produce downregulation of iNOS, suggesting that another uncharacterized event(s) is involved in this pathway. Dissection of the steps involved in the switch from the EBV latent cycle to the lytic cycle will be important to understand how virus-host relationships modulate the innate immune system.
Figures


Similar articles
-
Phosphatidylinositol 3-kinase/Akt pathway targets acetylation of Smad3 through Smad3/CREB-binding protein interaction: contribution to transforming growth factor beta1-induced Epstein-Barr virus reactivation.J Biol Chem. 2009 Sep 4;284(36):23912-24. doi: 10.1074/jbc.M109.036483. Epub 2009 Jul 9. J Biol Chem. 2009. PMID: 19589780 Free PMC article.
-
Repression of Smad-dependent transforming growth factor-beta signaling by Epstein-Barr virus latent membrane protein 1 through nuclear factor-kappaB.Int J Cancer. 2003 Jul 10;105(5):661-8. doi: 10.1002/ijc.11146. Int J Cancer. 2003. PMID: 12740915
-
Hemozoin increases IFN-gamma-inducible macrophage nitric oxide generation through extracellular signal-regulated kinase- and NF-kappa B-dependent pathways.J Immunol. 2003 Oct 15;171(8):4243-53. doi: 10.4049/jimmunol.171.8.4243. J Immunol. 2003. PMID: 14530348
-
Transforming growth factor beta 1 stimulates expression of the Epstein-Barr virus BZLF1 immediate-early gene product ZEBRA by an indirect mechanism which requires the MAPK kinase pathway.J Virol. 2000 Jul;74(13):5810-8. doi: 10.1128/jvi.74.13.5810-5818.2000. J Virol. 2000. PMID: 10846060 Free PMC article.
-
Lytic cycle switches of oncogenic human gammaherpesviruses.Adv Cancer Res. 2007;97:81-109. doi: 10.1016/S0065-230X(06)97004-3. Adv Cancer Res. 2007. PMID: 17419942 Review.
Cited by
-
Protective role of propofol on the kidney during early unilateral ureteral obstruction through inhibition of epithelial-mesenchymal transition.Am J Transl Res. 2016 Feb 15;8(2):460-72. eCollection 2016. Am J Transl Res. 2016. PMID: 27158339 Free PMC article.
-
Cysteine Alkylation in Enzymes and Transcription Factors: A Therapeutic Strategy for Cancer.Cancers (Basel). 2025 Jun 3;17(11):1876. doi: 10.3390/cancers17111876. Cancers (Basel). 2025. PMID: 40507356 Free PMC article. Review.
-
Over-production of nitric oxide by oxidative stress-induced activation of the TGF-β1/PI3K/Akt pathway in mesangial cells cultured in high glucose.Acta Pharmacol Sin. 2013 Apr;34(4):507-14. doi: 10.1038/aps.2012.207. Epub 2013 Mar 25. Acta Pharmacol Sin. 2013. PMID: 23524565 Free PMC article.
-
The epinephrine-induced PGE2 reduces Na+/K+ ATPase activity in Caco-2 cells via PKC, NF-κB and NO.PLoS One. 2019 Aug 8;14(8):e0220987. doi: 10.1371/journal.pone.0220987. eCollection 2019. PLoS One. 2019. PMID: 31393950 Free PMC article.
-
Protein kinase inhibitors that inhibit induction of lytic program and replication of Epstein-Barr virus.Antiviral Res. 2012 Dec;96(3):296-304. doi: 10.1016/j.antiviral.2012.09.021. Epub 2012 Oct 8. Antiviral Res. 2012. PMID: 23058855 Free PMC article.
References
-
- Agresti A., Lupo R., Bianchi M. E., Muller S. 2003. HMGB1 interacts differentially with members of the Rel family of transcription factors. Biochem. Biophys. Res. Commun. 302:421–426 - PubMed
-
- Arsura M., et al. 2003. Transient activation of NF-kappaB through a TAK1/IKK kinase pathway by TGF-beta1 inhibits AP-1/SMAD signaling and apoptosis: implications in liver tumor formation. Oncogene 22:412–425 - PubMed
-
- Bierie B., Moses H. L. 2006. Tumour microenvironment: TGFbeta: the molecular Jekyll and Hyde of cancer. Nat. Rev. Cancer 6:506–520 - PubMed
-
- Bonavida B., Baritaki S. 2011. Dual role of NO donors in the reversal of tumor cell resistance and EMT: Downregulation of the NF-kappaB/Snail/YY1/RKIP circuitry. Nitric Oxide 24:1–11 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

