Antibodies recognizing protective pertussis toxin epitopes are preferentially elicited by natural infection versus acellular immunization
- PMID: 21508166
- PMCID: PMC3122608
- DOI: 10.1128/CVI.00561-10
Antibodies recognizing protective pertussis toxin epitopes are preferentially elicited by natural infection versus acellular immunization
Abstract
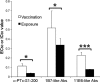
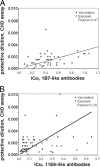
Despite more than 50 years of vaccination, disease caused by the bacterium Bordetella pertussis persists, with rates increasing in industrialized countries over the past decade. This rise may be attributed to several factors, including increased surveillance, emergence of vaccine escape variants, waning immunity in adults, and the introduction of acellular subunit vaccines, which include chemically detoxified pertussis toxin (PTd). Two potently protective epitopes on pertussis toxin (PTx) are recognized by the monoclonal antibodies 1B7 and 11E6, which inhibit catalytic and cell-binding activities, respectively. In order to determine whether the PTx exposure route affects antibody responses to these epitopes, we analyzed sera from 30 adults with confirmed pertussis exposure and from 30 recently vaccinated adults for specific anti-PTx antibody responses and in vitro CHO cell neutralization titers. While overall titers against PTx and the genetically detoxified variant, PTg, containing the R9K and E129G substitutions, were similar in the two groups, titers against specific epitopes depended on the exposure route. Natural infection resulted in significantly higher titers of anti-PTx-subunit 1, 1B7-like, and 11E6-like antibodies, while acellular vaccination resulted in significantly higher titers of antibodies recognizing PTd. We also observed a correlation between in vitro protection and the presence of 1B7-like and 11E6-like antibodies. Notably, chemical detoxification, as opposed to genetic inactivation, alters the PTx tertiary and quaternary structure, thereby affecting conformational epitopes and recognition of PTx by 1B7 and 11E6. The lower levels of serum antibodies recognizing clinically relevant epitopes after vaccination with PTd support inclusion of PTg in future vaccines.
Figures




References
-
- Anonymous 2007. Outbreaks of respiratory illness mistakenly attributed to pertussis—-New Hampshire, Massachusetts, and Tennessee, 2004-2006. MMWR Morb. Mortal. Wkly. Rep. 56:837–842 - PubMed
-
- Bartoloni A., et al. 1988. Mapping of a protective epitope of pertussis toxin by in vitro refolding of recombinant fragments. Nat. Biotechnol. 6:709–712
-
- Broder K. R., et al. 2006. Preventing tetanus, diphtheria, and pertussis among adolescents: use of tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccines recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm. Rep. 55:1–34 - PubMed
-
- Bruss J. B., et al. 1999. Treatment of severe pertussis: a study of the safety and pharmacology of intravenous pertussis immunoglobulin. Pediatr. Infect. Dis. J. 18:505–511 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

