Desmosomes: new perpetrators in tumour suppression
- PMID: 21508970
- PMCID: PMC3799918
- DOI: 10.1038/nrc3051
Desmosomes: new perpetrators in tumour suppression
Abstract
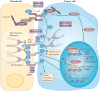
Adherens junctions, which are intercellular adhesive complexes that are crucial for maintaining epithelial homeostasis, are downregulated in many cancers to promote tumour progression. However, the role of desmosomes - adhesion complexes that are related to adherens junctions - in carcinogenesis has remained elusive. Recent studies using mouse genetic approaches have uncovered a role for desmosomes in tumour suppression, demonstrating that desmosome downregulation occurs before that of adherens junctions to drive tumour development and early invasion, suggesting a two-step model of adhesion dysfunction in cancer progression.
Figures


Similar articles
-
Adherens Junctions and Desmosomes Coordinate Mechanics and Signaling to Orchestrate Tissue Morphogenesis and Function: An Evolutionary Perspective.Cold Spring Harb Perspect Biol. 2018 Nov 1;10(11):a029207. doi: 10.1101/cshperspect.a029207. Cold Spring Harb Perspect Biol. 2018. PMID: 28893859 Free PMC article. Review.
-
Zipper-like series of desmosomes supported by subplasmalemmal actin belts in thymic epithelial reticular cells in the rat.Ann Anat. 2013 Jul;195(4):359-364. doi: 10.1016/j.aanat.2013.02.007. Epub 2013 Mar 18. Ann Anat. 2013. PMID: 23583310
-
Cell-Cell Junctions Organize Structural and Signaling Networks.Cold Spring Harb Perspect Biol. 2018 Apr 2;10(4):a029181. doi: 10.1101/cshperspect.a029181. Cold Spring Harb Perspect Biol. 2018. PMID: 28600395 Free PMC article. Review.
-
In vivo function of desmosomes.J Dermatol. 2004 Mar;31(3):171-87. doi: 10.1111/j.1346-8138.2004.tb00654.x. J Dermatol. 2004. PMID: 15187337 Review.
-
Junctional complexes in the early mammalian embryo.Semin Reprod Med. 2000;18(2):185-93. doi: 10.1055/s-2000-12557. Semin Reprod Med. 2000. PMID: 11256168 Review.
Cited by
-
Desmoglein 3: a help or a hindrance in cancer progression?Cancers (Basel). 2015 Jan 26;7(1):266-86. doi: 10.3390/cancers7010266. Cancers (Basel). 2015. PMID: 25629808 Free PMC article. Review.
-
Transmembrane protein PERP is a component of tessellate junctions and of other junctional and non-junctional plasma membrane regions in diverse epithelial and epithelium-derived cells.Cell Tissue Res. 2013 Jul;353(1):99-115. doi: 10.1007/s00441-013-1645-3. Epub 2013 May 21. Cell Tissue Res. 2013. PMID: 23689684 Free PMC article.
-
Galectin-3 regulates desmoglein-2 and intestinal epithelial intercellular adhesion.J Biol Chem. 2014 Apr 11;289(15):10510-10517. doi: 10.1074/jbc.M113.538538. Epub 2014 Feb 24. J Biol Chem. 2014. PMID: 24567334 Free PMC article.
-
Restoration of desmosomal junction protein expression and inhibition of H3K9-specific histone demethylase activity by cytostatic proline-rich polypeptide-1 leads to suppression of tumorigenic potential in human chondrosarcoma cells.Mol Clin Oncol. 2015 Jan;3(1):171-178. doi: 10.3892/mco.2014.445. Epub 2014 Oct 16. Mol Clin Oncol. 2015. PMID: 25469290 Free PMC article.
-
Down-regulated γ-catenin expression is associated with tumor aggressiveness in esophageal cancer.World J Gastroenterol. 2014 May 21;20(19):5839-48. doi: 10.3748/wjg.v20.i19.5839. World J Gastroenterol. 2014. PMID: 24914344 Free PMC article.
References
-
- Cooper GM. Oncogenes. Jones and Barlett Publishers; Boston: 1995.
-
- Schock F, Perrimon N. Molecular mechanisms of epithelial morphogenesis. Annu Rev Cell Dev Biol. 2002;18:463–493. - PubMed
-
- Jamora C, Fuchs E. Intercellular adhesion, signalling and the cytoskeleton. Nature Cell Biol. 2002;4:e101–e108. - PubMed
-
- Simpson CL, Green KJ. Desmosomes: new perspectives on a classic. J Invest Dermatol. 2007;127:2499–2515. - PubMed
-
- Vasioukhin V, Bauer C, Yin M, Fuchs E. Directed actin polymerization is the driving force for epithelial cell-cell adhesion. Cell. 2000;100:209–219. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

