Nucleolar disruption and apoptosis are distinct neuronal responses to etoposide-induced DNA damage
- PMID: 21517844
- PMCID: PMC3107349
- DOI: 10.1111/j.1471-4159.2011.07279.x
Nucleolar disruption and apoptosis are distinct neuronal responses to etoposide-induced DNA damage
Abstract
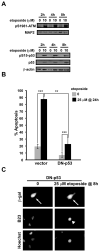
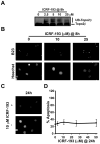
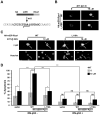
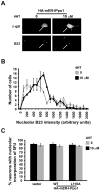
Although DNA damaging topoisomerase inhibitors induce apoptosis in developing neurons, their effects on adult neurons have not yet been characterized. We report a blockage of RNA-Polymerase-1-driven transcription and nucleolar stress in neocortical neurons of adult rats after intracarotid injection of the DNA-topoisomerase-2 inhibitor, etoposide. Intracerebroventricular injection of etoposide induced a similar response in neonatal rats. In contrast, etoposide triggered neuronal apoptosis in the neonates, but not the adults. Nucleolar disruption and apoptosis were also observed in etoposide-challenged cultured cortical neurons from newborn rats. In that system, activation of the DNA double strand break signaling kinase ataxia telangiectasia-mutated protein kinase, p53 and p53-dependent apoptosis required lower etoposide concentrations than did the p53-independent induction of nucleolar stress. These distinct responses may be coupled to different forms of etoposide-induced DNA damage. Indeed, double strand breaks by the over-expressed endonuclease I-Ppo1 were sufficient to induce p53-dependent apoptosis. Moreover, nucleolar transcription was insensitive to such damage implying single strand breaks and/or topoisomerase-2-DNA adducts as triggers of nucleolar stress. Because nucleolar stress is not age-restricted, it may underlie non-apoptotic neurotoxicity of chemotherapy- or neurodegeneration-associated DNA damage by reducing ribosomal biogenesis in adult brain. Conversely, nucleolar insensitivity to double strand breaks likely contributes to mature neuron tolerance of such lesions.
© 2011 The Authors. Journal of Neurochemistry © 2011 International Society for Neurochemistry.
Figures






Similar articles
-
Inhibitors of histone deacetylases enhance neurotoxicity of DNA damage.Neuromolecular Med. 2014 Dec;16(4):727-41. doi: 10.1007/s12017-014-8322-x. Epub 2014 Jul 26. Neuromolecular Med. 2014. PMID: 25063076 Free PMC article.
-
Proapoptotic Requirement of Ribosomal Protein L11 in Ribosomal Stress-Challenged Cortical Neurons.Mol Neurobiol. 2018 Jan;55(1):538-553. doi: 10.1007/s12035-016-0336-y. Epub 2016 Dec 14. Mol Neurobiol. 2018. PMID: 27975169 Free PMC article.
-
Inhibition of ATM blocks the etoposide-induced DNA damage response and apoptosis of resting human T cells.DNA Repair (Amst). 2012 Nov 1;11(11):864-73. doi: 10.1016/j.dnarep.2012.08.006. Epub 2012 Oct 9. DNA Repair (Amst). 2012. PMID: 23058634
-
Cytometry of ATM activation and histone H2AX phosphorylation to estimate extent of DNA damage induced by exogenous agents.Cytometry A. 2007 Sep;71(9):648-61. doi: 10.1002/cyto.a.20426. Cytometry A. 2007. PMID: 17622968 Free PMC article. Review.
-
Ku, Artemis, and ataxia-telangiectasia-mutated: signalling networks in DNA damage.Int J Biochem Cell Biol. 2008;40(4):598-603. doi: 10.1016/j.biocel.2007.12.007. Epub 2007 Dec 24. Int J Biochem Cell Biol. 2008. PMID: 18243767 Review.
Cited by
-
Up-regulation of Smurf1 after spinal cord injury in adult rats.J Mol Histol. 2013 Aug;44(4):381-90. doi: 10.1007/s10735-013-9499-2. Epub 2013 Apr 18. J Mol Histol. 2013. PMID: 23595775
-
Inhibitors of histone deacetylases enhance neurotoxicity of DNA damage.Neuromolecular Med. 2014 Dec;16(4):727-41. doi: 10.1007/s12017-014-8322-x. Epub 2014 Jul 26. Neuromolecular Med. 2014. PMID: 25063076 Free PMC article.
-
Ribosomal DNA and Neurological Disorders.Curr Mol Med. 2025;25(5):556-566. doi: 10.2174/0115665240292079240513093708. Curr Mol Med. 2025. PMID: 38778614 Review.
-
Dynamic crosstalk between amino acid metabolism and cancer drug efficacy: From mechanisms to therapeutic opportunities.iScience. 2025 Apr 11;28(5):112405. doi: 10.1016/j.isci.2025.112405. eCollection 2025 May 16. iScience. 2025. PMID: 40625405 Free PMC article. Review.
-
Downregulation of miR-23a and miR-27a following experimental traumatic brain injury induces neuronal cell death through activation of proapoptotic Bcl-2 proteins.J Neurosci. 2014 Jul 23;34(30):10055-71. doi: 10.1523/JNEUROSCI.1260-14.2014. J Neurosci. 2014. PMID: 25057207 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

