Origins of ion selectivity in potassium channels from the perspective of channel block
- PMID: 21518829
- PMCID: PMC3082928
- DOI: 10.1085/jgp.201010551
Origins of ion selectivity in potassium channels from the perspective of channel block
Figures
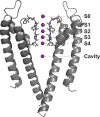
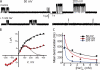
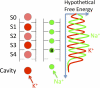
Similar articles
-
A minimalist model for ion partitioning and competition in a K+ channel selectivity filter.J Gen Physiol. 2011 Sep;138(3):371-3. doi: 10.1085/jgp.201110694. J Gen Physiol. 2011. PMID: 21875982 Free PMC article. No abstract available.
-
Structural studies of ion selectivity in tetrameric cation channels.J Gen Physiol. 2011 May;137(5):397-403. doi: 10.1085/jgp.201010546. J Gen Physiol. 2011. PMID: 21518828 Free PMC article. Review. No abstract available.
-
Structure of potassium channels.Cell Mol Life Sci. 2015 Oct;72(19):3677-93. doi: 10.1007/s00018-015-1948-5. Epub 2015 Jun 13. Cell Mol Life Sci. 2015. PMID: 26070303 Free PMC article. Review.
-
Pore hydration states of KcsA potassium channels in membranes.J Biol Chem. 2015 Oct 30;290(44):26765-75. doi: 10.1074/jbc.M115.661819. Epub 2015 Sep 14. J Biol Chem. 2015. PMID: 26370089 Free PMC article.
-
Crystallographic study of the tetrabutylammonium block to the KcsA K+ channel.J Mol Biol. 2007 Feb 23;366(3):806-14. doi: 10.1016/j.jmb.2006.11.081. Epub 2006 Dec 2. J Mol Biol. 2007. PMID: 17196615
Cited by
-
Ion behavior in the selectivity filter of HCN1 channels.Biophys J. 2022 Jun 7;121(11):2206-2218. doi: 10.1016/j.bpj.2022.04.024. Epub 2022 Apr 26. Biophys J. 2022. PMID: 35474263 Free PMC article.
-
Optimal transport and colossal ionic mechano-conductance in graphene crown ethers.Sci Adv. 2019 Jul 12;5(7):eaaw5478. doi: 10.1126/sciadv.aaw5478. eCollection 2019 Jul. Sci Adv. 2019. PMID: 31309155 Free PMC article.
-
Structural properties determining low K+ affinity of the selectivity filter in the TWIK1 K+ channel.J Biol Chem. 2018 May 4;293(18):6969-6984. doi: 10.1074/jbc.RA118.001817. Epub 2018 Mar 15. J Biol Chem. 2018. PMID: 29545310 Free PMC article.
-
Multi-ion free energy landscapes underscore the microscopic mechanism of ion selectivity in the KcsA channel.Biochim Biophys Acta. 2016 Jul;1858(7 Pt B):1722-32. doi: 10.1016/j.bbamem.2016.02.019. Epub 2016 Feb 16. Biochim Biophys Acta. 2016. PMID: 26896693 Free PMC article.
-
On the selective ion binding hypothesis for potassium channels.Proc Natl Acad Sci U S A. 2011 Nov 1;108(44):17963-8. doi: 10.1073/pnas.1110735108. Epub 2011 Oct 19. Proc Natl Acad Sci U S A. 2011. PMID: 22011574 Free PMC article.
References
-
- Alberts B., Johnson A., Lewis J., Raff M., Roberts K., Walter P. 2007. Molecular Biology of the Cell. Fifth edition Garland Science, New York: 1392 pp
-
- Allen T.W., Hoyles M., Kuyucak S., Chung S.H. 1999. Molecular and Brownian dynamics study of ion selectivity and conductivity in the potassium channel. Chem. Phys. Lett. 313:358–365 10.1016/S0009-2614(99)01004-0 - DOI
-
- Allen T.W., Bliznyuk A., Rendell A.P., Kuyucak S., Chung S.H. 2000. The potassium channel: structure, selectivity and diffusion. J. Chem. Phys. 112:8191–8204 10.1063/1.481420 - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

