Principles of planar polarity in animal development
- PMID: 21521735
- PMCID: PMC3082295
- DOI: 10.1242/dev.054080
Principles of planar polarity in animal development
Abstract
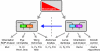
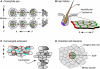
Planar polarity describes the coordinated polarisation of cells or structures in the plane of a tissue. The patterning mechanisms that underlie planar polarity are well characterised in Drosophila, where many events are regulated by two pathways: the 'core' planar polarity complex and the Fat/Dachsous system. Components of both pathways also function in vertebrates and are implicated in diverse morphogenetic processes, some of which self-evidently involve planar polarisation and some of which do not. Here, we review the molecular mechanisms and cellular consequences of planar polarisation in diverse contexts, seeking to identify the common principles across the animal kingdom.
Figures


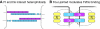



References
-
- Adler P. N. (1992). The genetic control of tissue polarity in Drosophila. BioEssays 14, 735-741 - PubMed
-
- Adler P. N. (2002). Planar signalling and morphogenesis in Drosophila. Dev. Cell 2, 525-535 - PubMed
-
- Adler P. N., Krasnow R. E., Liu J. (1997). Tissue polarity points from cells that have higher Frizzled levels towards cells that have lower Frizzled levels. Curr. Biol. 7, 940-949 - PubMed
-
- Adler P., Charlton J., Liu J. (1998). Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype by altering frizzled signaling. Development 125, 959-968 - PubMed
-
- Adler P. N., Liu J., Charlton J. (2000). Cell size and the morphogenesis of wing hairs in Drosophila. Genesis 28, 82-91 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

