Molecular profiling of a lethal tumor microenvironment, as defined by stromal caveolin-1 status in breast cancers
- PMID: 21521946
- PMCID: PMC3142463
- DOI: 10.4161/cc.10.11.15675
Molecular profiling of a lethal tumor microenvironment, as defined by stromal caveolin-1 status in breast cancers
Abstract
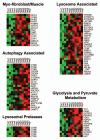
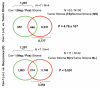
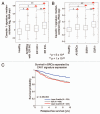
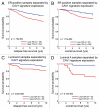
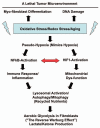
Breast cancer progression and metastasis are driven by complex and reciprocal interactions, between epithelial cancer cells and their surrounding stromal microenvironment. We have previously shown that a loss of stromal Cav-1 expression is associated with an increased risk of early tumor recurrence, metastasis and decreased overall survival. To identify and characterize the signaling pathways that are activated in Cav-1 negative tumor stroma, we performed gene expression profiling using laser microdissected breast cancer-associated stroma. Tumor stroma was laser capture microdissected from 4 cases showing high stromal Cav-1 expression and 7 cases with loss of stromal Cav-1. Briefly, we identified 238 gene transcripts that were upregulated and 232 gene transcripts that were downregulated in the stroma of tumors showing a loss of Cav-1 expression (p ≤ 0.01 and fold-change ≥ 1.5). Gene set enrichment analysis (GSEA) revealed "stemness," inflammation, DNA damage, aging, oxidative stress, hypoxia, autophagy and mitochondrial dysfunction in the tumor stroma of patients lacking stromal Cav-1. Our findings are consistent with the recently proposed "Reverse Warburg Effect" and the "Autophagic Tumor Stroma Model of Cancer Metabolism." In these two complementary models, cancer cells induce oxidative stress in adjacent stromal cells, which then forces these stromal fibroblasts to undergo autophagy/mitophagy and aerobic glycolysis. This, in turn, produces recycled nutrients (lactate, ketones and glutamine) to feed anabolic cancer cells, which are undergoing oxidative mitochondrial metabolism. Our results are also consistent with previous biomarker studies showing that the increased expression of known autophagy markers (such as ATG16L and the cathepsins) in the tumor stroma is specifically associated with metastatic tumor progression and/or poor clinical outcome.
Figures







Comment in
-
Defining bad stroma in human breast tumors.Cell Cycle. 2011 Sep 15;10(18):3056. doi: 10.4161/cc.10.18.17047. Epub 2011 Sep 15. Cell Cycle. 2011. PMID: 21912214 No abstract available.
-
Caveolin-1: would-be Achilles' heel of tumor microenvironment?Cell Cycle. 2011 Oct 15;10(20):3431. doi: 10.4161/cc.10.20.17648. Epub 2011 Oct 15. Cell Cycle. 2011. PMID: 22030625 No abstract available.
References
-
- Witkiewicz AK, Casimiro MC, Dasgupta A, Mercier I, Wang C, Bonuccelli G, et al. Towards a new “stromal-based” classification system for human breast cancer prognosis and therapy. Cell Cycle. 2009;8:1654–1658. - PubMed
-
- Witkiewicz AK, Dasgupta A, Nguyen KH, Liu C, Kovatich AJ, Schwartz GF, et al. Stromal caveolin-1 levels predict early DCIS progression to invasive breast cancer. Cancer Biol Ther. 2009;8:1167–1175. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
