Conductive AFM patterning on oligo(ethylene glycol)-terminated alkyl monolayers on silicon substrates: proposed mechanism and fabrication of avidin patterns
- PMID: 21526810
- PMCID: PMC3230272
- DOI: 10.1021/la1047358
Conductive AFM patterning on oligo(ethylene glycol)-terminated alkyl monolayers on silicon substrates: proposed mechanism and fabrication of avidin patterns
Abstract
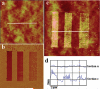
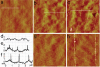
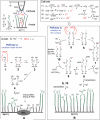
Micro- and nanopatterns of biomolecules on inert, ultrathin platforms on nonoxidized silicon are ideal interfaces between silicon-based microelectronics and biological systems. We report here the local oxidation nanolithography with conductive atomic force microscopy (cAFM) on highly protein-resistant, oligo(ethylene glycol) (OEG)-terminated alkyl monolayers on nonoxidized silicon substrates. We propose a mechanism for this process, suggesting that it is possible to oxidize only the top ethylene glycol units to generate carboxylic acid and aldehyde groups on the film surface. We show that avidin molecules can be attached selectively to the oxidized pattern and the density can be varied by altering the bias voltage during cAFM patterning. Biotinylated molecules and nanoparticles are selectively immobilized on the resultant avidin patterns. Since one of the most established methods for immobilization of biomolecules is based on avidin-biotin binding and a wide variety of biotinylated biomolecules are available, this approach represents a versatile means for prototyping any nanostructures presenting these biomolecules on silicon substrates.
Figures


Similar articles
-
Nanometric protein arrays on protein-resistant monolayers on silicon surfaces.J Am Chem Soc. 2004 Jul 7;126(26):8098-9. doi: 10.1021/ja048405x. J Am Chem Soc. 2004. PMID: 15225034
-
Micropatterned avidin arrays on silicon substrates via photolithography, self-assembly and bioconjugation.Biotechnol Appl Biochem. 2006 Feb;43(Pt 2):85-91. doi: 10.1042/BA20050085. Biotechnol Appl Biochem. 2006. PMID: 16218906
-
Charge patterns as templates for the assembly of layered biomolecular structures.J Nanosci Nanotechnol. 2006 Aug;6(8):2445-50. doi: 10.1166/jnn.2006.516. J Nanosci Nanotechnol. 2006. PMID: 17037854
-
Monolayer-derivative functionalization of non-oxidized silicon surfaces.Chem Rec. 2005;5(3):145-59. doi: 10.1002/tcr.20041. Chem Rec. 2005. PMID: 15889408 Review.
-
Lab on a tip: Applications of functional atomic force microscopy for the study of electrical properties in biology.Acta Biomater. 2019 Nov;99:33-52. doi: 10.1016/j.actbio.2019.08.023. Epub 2019 Aug 16. Acta Biomater. 2019. PMID: 31425893 Review.
Cited by
-
Spatially selective surface platforms for binding fibrinogen prepared by particle lithography with organosilanes.Interface Focus. 2013 Jun 6;3(3):20120102. doi: 10.1098/rsfs.2012.0102. Interface Focus. 2013. PMID: 24427541 Free PMC article.
-
Anisotropic Effects in Local Anodic Oxidation Nanolithography on Silicon Surfaces: Insights from ReaxFF Molecular Dynamics.Langmuir. 2024 Jul 15;40(30):15530-40. doi: 10.1021/acs.langmuir.4c01129. Online ahead of print. Langmuir. 2024. PMID: 39008811 Free PMC article.
References
-
- Ahmad SAA, Wong LS, Ul-Haq E, Hobbs JK, Leggett GJ, Micklefield J. J. Am. Chem. Soc. 2009;131:1513. - PubMed
- Arnold M, Cavalcanti-Adam EA, Glass R, Blummel J, Eck W, Kantlehner M, Kessler H, Spatz JP. ChemPhysChem. 2004;5:383. - PubMed
- Ballav N, Thomas H, Winkler T, Terfort A, Zharnikov M. Angew. Chem., Int. Ed. 2009;48:5833. - PubMed
- Christman KL, Enriquez-Rios VD, Maynard HD. Soft Matter. 2006;2:928. - PubMed
- Jonkheijm P, Weinrich D, Schroder H, Niemeyer CM, Waldmann H. Angew. Chem., Int. Ed. 2008;47:9618. - PubMed
- Ngunjiri J, Garno JC. Anal. Chem. 2008;80:1361. - PubMed
- Ogaki R, Alexander M, Kingshott P. Mater. Today. 2010;13:22.
- Saavedra HM, Mullen TJ, Zhang PP, Dewey DC, Claridge SA, Weiss PS. Rep. Prog. Phys. 2010;73:40.
-
- Ducker R, Garcia A, Zhang JM, Chen T, Zauscher S. Soft Matter. 2008;4:1774.
-
- Salaita K, Wang YH, Mirkin CA. Nat. Nanotechnol. 2007;2:145. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

