Cetuximab plus irinotecan in pretreated metastatic colorectal cancer patients: the ELSIE study
- PMID: 21528063
- PMCID: PMC3080724
- DOI: 10.3748/wjg.v17.i14.1879
Cetuximab plus irinotecan in pretreated metastatic colorectal cancer patients: the ELSIE study
Abstract
Aim: To evaluate the efficacy and safety of cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer (mCRC) patients from South-East Asia and Australia.
Methods: In this open-label, phase II study, the main eligibility criteria were epidermal growth factor receptor-positive mCRC with progressive disease within 3 mo of an irinotecan-based regimen as the most recent chemotherapy. Patients received cetuximab 400 mg/m2 initially, then 250 mg/m2 every week, with the same regimen of irinotecan on which the patients had progressed (4 pre-defined regimens allowed). The primary objective was evaluation of progression-free survival (PFS) at 12 wk. Secondary objectives included a further investigation of PFS, and an assessment of the overall response rate (ORR), duration of response, time to treatment failure (TTF), overall survival and the safety profile.
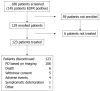
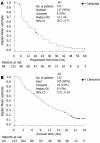
Results: One hundred and twenty nine patients were enrolled from 25 centers in the Asia-Pacific region and of these 123 received cetuximab plus irinotecan. The most common recent irinotecan regimen used was 180 mg/m2 every 2 wk which had been used in 93 patients (75.6%). The PFS rate at 12 wk was 50% (95% confidence interval (CI, 41-59) and median PFS time was 12.1 wk (95% CI: 9.7-17.7). The ORR was 13.8% (95% CI: 8.3-21.2) and disease control rate was 49.6% (95% CI: 40.5-58.8). Median duration of response was 31.1 wk (95% CI: 18.0-42.6) and median overall survival was 9.5 mo (95% CI, 7.5-11.7). The median TTF was 11.7 wk (95% CI: 9.1-17.4). Treatment was generally well tolerated. The most common grade 3/4 adverse events were diarrhea (13.8%), neutropenia (8.9%), rash (5.7%) and vomiting (5.7%).
Conclusion: In patients from Asia and Australia, this study confirms the activity and safety of cetuximab plus irinotecan observed in previous studies in Europe and South America.
Keywords: Asia; Cetuximab; Epidermal growth factor receptor; Irinotecan; Metastatic colorectal cancer.
Figures
References
-
- Ferlay J, Bray F, Pisani P, Parkin DM. GLOBOCAN. Cancer incidence, mortality and prevalence worldwide, vol. Version 1.0, 2004: IARC Press, 2002
-
- Van Cutsem E, Nordlinger B, Adam R, Köhne CH, Pozzo C, Poston G, Ychou M, Rougier P. Towards a pan-European consensus on the treatment of patients with colorectal liver metastases. Eur J Cancer. 2006;42:2212–2221. - PubMed
-
- Center MM, Jemal A, Ward E. International trends in colorectal cancer incidence rates. Cancer Epidemiol Biomarkers Prev. 2009;18:1688–1694. - PubMed
-
- Sung JJ, Lau JY, Goh KL, Leung WK. Increasing incidence of colorectal cancer in Asia: implications for screening. Lancet Oncol. 2005;6:871–876. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials