Raphe serotonin neurons are not homogenous: electrophysiological, morphological and neurochemical evidence
- PMID: 21530552
- PMCID: PMC3120045
- DOI: 10.1016/j.neuropharm.2011.04.008
Raphe serotonin neurons are not homogenous: electrophysiological, morphological and neurochemical evidence
Abstract
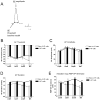
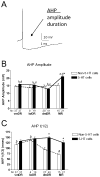
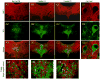
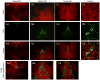
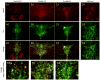
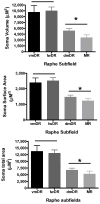
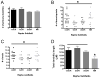
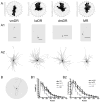
The median (MR) and dorsal raphe (DR) nuclei contain the majority of the 5-hydroxytryptamine (5-HT, serotonin) neurons that project to limbic forebrain regions, are important in regulating homeostatic functions and are implicated in the etiology and treatment of mood disorders and schizophrenia. The primary synaptic inputs within and to the raphe are glutamatergic and GABAergic. The DR is divided into three subfields, i.e., ventromedial (vmDR), lateral wings (lwDR) and dorsomedial (dmDR). Our previous work shows that cell characteristics of 5-HT neurons and the magnitude of the 5-HT(1A) and 5-HT(1B) receptor-mediated responses in the vmDR and MR are not the same. We extend these observations to examine the electrophysiological properties across all four raphe subfields in both 5-HT and non-5-HT neurons. The neurochemical topography of glutamatergic and GABAergic cell bodies and nerve terminals were identified using immunohistochemistry and the morphology of the 5-HT neurons was measured. Although 5-HT neurons possessed similar physiological properties, important differences existed between subfields. Non-5-HT neurons were indistinguishable from 5-HT neurons. GABA neurons were distributed throughout the raphe, usually in areas devoid of 5-HT neurons. Although GABAergic synaptic innervation was dense throughout the raphe (immunohistochemical analysis of the GABA transporters GAT1 and GAT3), their distributions differed. Glutamate neurons, as defined by vGlut3 anti-bodies, were intermixed and co-localized with 5-HT neurons within all raphe subfields. Finally, the dendritic arbor of the 5-HT neurons was distinct between subfields. Previous studies regard 5-HT neurons as a homogenous population. Our data support a model of the raphe as an area composed of functionally distinct subpopulations of 5-HT and non-5-HT neurons, in part delineated by subfield. Understanding the interaction of the cell properties of the neurons in concert with their morphology, local distribution of GABA and glutamate neurons and their synaptic input, reveals a more complicated and heterogeneous raphe. These results provide an important foundation for understanding how specific subfields modulate behavior and for defining which aspects of the circuitry are altered during the etiology of psychological disorders.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures








References
-
- Abrams JK, Johnson PL, Hay-Schmidt A, Mikkelsen JD, Shekhar A, Lowry CA. Serotonergic systems associated with arousal and vigilance behaviors following administration of anxiogenic drugs. Neuroscience. 2005;133:983–997. - PubMed
-
- Abrams JK, Johnson PL, Hollis JH, Lowry CA. Anatomic and functional topography of the dorsal raphe nucleus. Annals of the New York Academy of Sciences. 2004;1018:46–57. - PubMed
-
- Adell A, Casanovas JM, Artigas F. Comparative study in the rat of the actions of different types of stress on the release of 5-HT in raphe nuclei and forebrain areas. Neuropharmacology. 1997;36:735–741. - PubMed
-
- Aghajanian GK, Lakoski JM. Hyperpolarization of serotonergic neurons by serotonin and LSD: studies in brain slices showing increased K+ conductance. Brain research. 1984;305:181–185. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

