Function and mechanism of axonal targeting of voltage-sensitive potassium channels
- PMID: 21530607
- PMCID: PMC3112463
- DOI: 10.1016/j.pneurobio.2011.04.009
Function and mechanism of axonal targeting of voltage-sensitive potassium channels
Abstract
Precise localization of various ion channels into proper subcellular compartments is crucial for neuronal excitability and synaptic transmission. Axonal K(+) channels that are activated by depolarization of the membrane potential participate in the repolarizing phase of the action potential, and hence regulate action potential firing patterns, which encode output signals. Moreover, some of these channels can directly control neurotransmitter release at axonal terminals by constraining local membrane excitability and limiting Ca(2+) influx. K(+) channels differ not only in biophysical and pharmacological properties, but in expression and subcellular distribution as well. Importantly, proper targeting of channel proteins is a prerequisite for electrical and chemical functions of axons. In this review, we first highlight recent studies that demonstrate different roles of axonal K(+) channels in the local regulation of axonal excitability. Next, we focus on research progress in identifying axonal targeting motifs and machinery of several different types of K(+) channels present in axons. Regulation of K(+) channel targeting and activity may underlie a novel form of neuronal plasticity. This research field can contribute to generating novel therapeutic strategies through manipulating neuronal excitability in treating neurological diseases, such as multiple sclerosis, neuropathic pain, and Alzheimer's disease.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
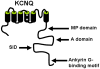
Kv7/KCNQ/M-channels in rat glutamatergic hippocampal axons and their role in regulation of excitability and transmitter release.J Physiol. 2006 Oct 1;576(Pt 1):235-56. doi: 10.1113/jphysiol.2006.111336. Epub 2006 Jul 13. J Physiol. 2006. PMID: 16840518 Free PMC article.
-
A conserved domain in axonal targeting of Kv1 (Shaker) voltage-gated potassium channels.Science. 2003 Aug 1;301(5633):646-9. doi: 10.1126/science.1086998. Science. 2003. PMID: 12893943
-
Distinct frequency-dependent regulation of nerve terminal excitability and synaptic transmission by IA and IK potassium channels revealed by Drosophila Shaker and Shab mutations.J Neurosci. 2006 Jun 7;26(23):6238-48. doi: 10.1523/JNEUROSCI.0862-06.2006. J Neurosci. 2006. PMID: 16763031 Free PMC article.
-
Diverse roles for auxiliary subunits in phosphorylation-dependent regulation of mammalian brain voltage-gated potassium channels.Pflugers Arch. 2011 Nov;462(5):631-43. doi: 10.1007/s00424-011-1004-8. Epub 2011 Aug 6. Pflugers Arch. 2011. PMID: 21822597 Free PMC article. Review.
-
Axonal voltage-gated ion channels as pharmacological targets for pain.Eur J Pharmacol. 2013 May 15;708(1-3):105-12. doi: 10.1016/j.ejphar.2013.03.001. Epub 2013 Mar 13. Eur J Pharmacol. 2013. PMID: 23500193 Review.
Cited by
-
Balanced Activity between Kv3 and Nav Channels Determines Fast-Spiking in Mammalian Central Neurons.iScience. 2018 Nov 30;9:120-137. doi: 10.1016/j.isci.2018.10.014. Epub 2018 Oct 18. iScience. 2018. PMID: 30390433 Free PMC article.
-
Suppression of Inflammatory Demyelinaton and Axon Degeneration through Inhibiting Kv3 Channels.Front Mol Neurosci. 2017 Oct 26;10:344. doi: 10.3389/fnmol.2017.00344. eCollection 2017. Front Mol Neurosci. 2017. PMID: 29123469 Free PMC article.
-
At the Fulcrum in Health and Disease: Cdk5 and the Balancing Acts of Neuronal Structure and Physiology.Brain Disord Ther. 2012 Jul;2012(Suppl 1):001. doi: 10.4172/2168-975X.S1-001. Brain Disord Ther. 2012. PMID: 25364642 Free PMC article.
-
Subcellular localization of K+ channels in mammalian brain neurons: remarkable precision in the midst of extraordinary complexity.Neuron. 2015 Jan 21;85(2):238-56. doi: 10.1016/j.neuron.2014.12.042. Neuron. 2015. PMID: 25611506 Free PMC article. Review.
-
Selective Loss of Presynaptic Potassium Channel Clusters at the Cerebellar Basket Cell Terminal Pinceau in Adam11 Mutants Reveals Their Role in Ephaptic Control of Purkinje Cell Firing.J Neurosci. 2015 Aug 12;35(32):11433-44. doi: 10.1523/JNEUROSCI.1346-15.2015. J Neurosci. 2015. PMID: 26269648 Free PMC article.
References
-
- Alioua A, Lu R, Kumar Y, Eghbali M, Kundu P, Toro L, Stefani E. Slo1 caveolin-binding motif, a mechanism of caveolin-1-Slo1 interaction regulating Slo1 surface expression. J Biol Chem. 2008;283:4808–4817. - PubMed
-
- Arnold DB, Clapham DE. Molecular determinants for subcellular localization of PSD-95 with an interacting K+ channel. Neuron. 1999;23:149–157. - PubMed
-
- Atzori M, Lau D, Tansey EP, Chow A, Ozaita A, Rudy B, McBain CJ. H2 histamine receptor-phosphorylation of Kv3.2 modulates interneuron fast spiking. Nat Neurosci. 2000;3:791–798. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

