Evolution of the amniote pallium and the origins of mammalian neocortex
- PMID: 21534989
- PMCID: PMC3384708
- DOI: 10.1111/j.1749-6632.2011.06006.x
Evolution of the amniote pallium and the origins of mammalian neocortex
Abstract
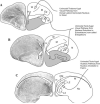
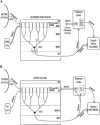
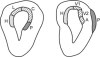
Karten's neocortex hypothesis holds that many component cell populations of the sauropsid dorsal ventricular ridge (DVR) are homologous to particular cell populations in layers of auditory and visual tectofugal-recipient neocortex of mammals (i.e., temporal neocortex), as well as to some amygdaloid populations. The claustroamygdalar hypothesis, based on gene expression domains, proposes that mammalian homologues of DVR are found in the claustrum, endopiriform nuclei, and/or pallial amygdala. Because hypotheses of homology need to account for the totality of the evidence, the available data on multiple forebrain features of sauropsids and mammals are reviewed here. While some genetic data are compatible with the claustroamygdalar hypothesis, and developmental (epigenetic) data are indecisive, hodological, morphological, and topographical data favor the neocortex hypothesis and are inconsistent with the claustroamygdalar hypothesis. Detailed studies of gene signaling cascades that establish neuronal cell-type identity in DVR, tectofugal-recipient neocortex, and claustroamygdala will be needed to resolve this debate about the evolution of neocortex.
© 2011 New York Academy of Sciences.
Figures

References
-
- Karten HJ. The organization of the avian telencephalon and some speculations on the phylogeny of the amniote telencephalon. In: Petras JN, Noback C, editors. Comparative and Evolutionary Aspects of the Vertebrate Central Nervous System. Vol. 167. Annals of the NewYork Academy of Science; New York: 1969. pp. 164–179.
-
- Karten HJ, Dubbeldam JL. The organization and projections of the paleostriatal complex in the pigeon (Columba livia). J. Comp. Neurol. 1973;148:61–89. - PubMed
-
- Karten HJ. The ascending auditory pathway in the pigeon (Columba livia): I. Diencephalic projections of the inferior colliculus (nucleus mesencephali lateralis, pars dorsalis). Brain Res. 1967;6:409–427. - PubMed
-
- Karten HJ. The ascending auditory pathway in the pigeon (Columba livia): II. Telencephalic projections of the nucleus ovoidalis thalami. Brain Res. 1968;11:134–153. - PubMed
-
- Karten HJ, Revzin AM. The afferent connections in the nucleus rotundus in the pigeon. Brain Res. 1966;2:368–377. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

