Tumor necrosis factor alpha production from CD8+ T cells mediates oviduct pathological sequelae following primary genital Chlamydia muridarum infection
- PMID: 21536799
- PMCID: PMC3191981
- DOI: 10.1128/IAI.05022-11
Tumor necrosis factor alpha production from CD8+ T cells mediates oviduct pathological sequelae following primary genital Chlamydia muridarum infection
Abstract
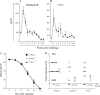
The immunopathogenesis of Chlamydia trachomatis-induced oviduct pathological sequelae is not well understood. Mice genetically deficient in perforin (perforin(-/-) mice) or tumor necrosis factor alpha (TNF-α) production (TNF-α(-/-) mice) displayed comparable vaginal chlamydial clearance rates but significantly reduced oviduct pathology (hydrosalpinx) compared to that of wild-type mice. Since both perforin and TNF-α are effector mechanisms of CD8(+) T cells, we evaluated the role of CD8(+) T cells during genital Chlamydia muridarum infection and oviduct sequelae. Following vaginal chlamydial challenge, (i) mice deficient in TAP I (and therefore the major histocompatibility complex [MHC] I pathway and CD8(+) T cells), (ii) wild-type mice depleted of CD8(+) T cells, and (iii) mice genetically deficient in CD8 (CD8(-/-) mice) all displayed similar levels of vaginal chlamydial clearance but significantly reduced hydrosalpinx, compared to those of wild-type C57BL/6 mice, suggesting a role for CD8(+) T cells in chlamydial pathogenesis. Repletion of CD8(-/-) mice with wild-type or perforin(-/-), but not TNF-α(-/-), CD8(+) T cells at the time of challenge restored hydrosalpinx to levels observed in wild-type C57BL/6 mice, suggesting that TNF-α production from CD8(+) T cells is important for pathogenesis. Additionally, repletion of TNF-α(-/-) mice with TNF-α(+/+) CD8(+) T cells significantly enhanced the incidence of hydrosalpinx and oviduct dilatation compared to those of TNF-α(-/-) mice but not to the levels found in wild-type mice, suggesting that TNF-α production from CD8(+) T cells and non-CD8(+) cells cooperates to induce optimal oviduct pathology following genital chlamydial infection. These results provide compelling new evidence supporting the contribution of CD8(+) T cells and TNF-α production to Chlamydia-induced reproductive tract sequelae.
Figures




References
-
- Alikhani M., Alikhani Z., Raptis M., Graves D. T. 2004. TNF-alpha in vivo stimulates apoptosis in fibroblasts through caspase-8 activation and modulates the expression of pro-apoptotic genes. J. Cell. Physiol. 201:341–348 - PubMed
-
- Bilenki L., et al. 2005. NK T cell activation promotes Chlamydia trachomatis infection in vivo. J. Immunol. 175:3197–3206 - PubMed
-
- Brunham R. C., Rey-Ladino J. 2005. Immunology of Chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nat. Rev. Immunol. 5:149–161 - PubMed
-
- Chatzidakis I., Mamalaki C. 2010. T cells as sources and targets of TNF: implications for immunity and autoimmunity. Curr. Dir. Autoimmun. 11:105–118 - PubMed
-
- Chen G., Goeddel D. V. 2002. TNF-R1 signaling: a beautiful pathway. Science 296:1634–1635 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

