Harnessing the potential of induced pluripotent stem cells for regenerative medicine
- PMID: 21540845
- PMCID: PMC3617981
- DOI: 10.1038/ncb0511-497
Harnessing the potential of induced pluripotent stem cells for regenerative medicine
Erratum in
- Nat Cell Biol. 2011 Jun;13(6):734
Abstract
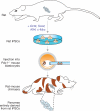
The discovery of methods to convert somatic cells into induced pluripotent stem cells (iPSCs) through expression of a small combination of transcription factors has raised the possibility of producing custom-tailored cells for the study and treatment of numerous diseases. Indeed, iPSCs have already been derived from patients suffering from a large variety of disorders. Here we review recent progress that has been made in establishing iPSC-based disease models, discuss associated technical and biological challenges, and highlight possible solutions to overcome these barriers. We believe that a better understanding of the molecular basis of pluripotency, cellular reprogramming and lineage-specific differentiation of iPSCs is necessary for progress in regenerative medicine.
Figures
References
-
- Gurdon JB. The developmental capacity of nuclei taken from intestinal epithelium cells of feeding tadpoles. J. Embryol. Exp. Morphol. 1962;10:622–640. - PubMed
-
- Rideout WM, III, Hochedlinger K, Kyba M, Daley GQ, Jaenisch R. Correction of a genetic defect by nuclear transplantation and combined cell and gene therapy. Cell. 2002;109:17–27. - PubMed
-
- Schnieke AE, et al. Human factor IX transgenic sheep produced by transfer of nuclei from transfected fetal fibroblasts. Science. 1997;278:2130–2133. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

