Differentiation generates paracrine cell pairs that maintain basaloid mouse mammary tumors: proof of concept
- PMID: 21541292
- PMCID: PMC3082567
- DOI: 10.1371/journal.pone.0019310
Differentiation generates paracrine cell pairs that maintain basaloid mouse mammary tumors: proof of concept
Abstract
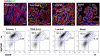
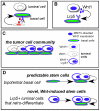
There is a paradox offered up by the cancer stem cell hypothesis. How are the mixed populations that are characteristic of heterogeneous solid tumors maintained at constant proportion, given their high, and different, mitotic indices? In this study, we evaluate a well-characterized mouse model of human basaloid tumors (induced by the oncogene Wnt1), which comprise mixed populations of mammary epithelial cells resembling their normal basal and luminal counterparts. We show that these cell types are substantially inter-dependent, since the MMTV LTR drives expression of Wnt1 ligand in luminal cells, whereas the functional Wnt1-responsive receptor (Lrp5) is expressed by basal cells, and both molecules are necessary for tumor growth. There is a robust tumor initiating activity (tumor stem cell) in the basal cell population, which is associated with the ability to differentiate into luminal and basal cells, to regenerate the oncogenic paracrine signaling cell pair. However, we found an additional tumor stem cell activity in the luminal cell population. Knowing that tumors depend upon Wnt1-Lrp5, we hypothesized that this stem cell must express Lrp5, and found that indeed, all the stem cell activity could be retrieved from the Lrp5-positive cell population. Interestingly, this reflects post-transcriptional acquisition of Lrp5 protein expression in luminal cells. Furthermore, this plasticity of molecular expression is reflected in plasticity of cell fate determination. Thus, in vitro, Wnt1-expressing luminal cells retro-differentiate to basal cell types, and in vivo, tumors initiated with pure luminal cells reconstitute a robust basal cell subpopulation that is indistinguishable from the populations initiated by pure basal cells. We propose this is an important proof of concept, demonstrating that bipotential tumor stem cells are essential in tumors where oncogenic ligand-receptor pairs are separated into different cell types, and suggesting that Wnt-induced molecular and fate plasticity can close paracrine loops that are usually separated into distinct cell types.
Conflict of interest statement
Figures
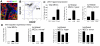


References
-
- Wicha MS, Liu S, Dontu G. Cancer stem cells: an old idea–a paradigm shift. Cancer Res. 2006;66:1883–1890; discussion 1895–1886. - PubMed
-
- Carey LA, Dees EC, Sawyer L, Gatti L, Moore DT, et al. The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res. 2007;13:2329–2334. - PubMed
-
- Badve S, Dabbs DJ, Schnitt SJ, Baehner FL, Decker T, et al. Basal-like and triple-negative breast cancers: a critical review with an emphasis on the implications for pathologists and oncologists. Mod Pathol 2010 - PubMed
-
- Rakha EA, Elsheikh SE, Aleskandarany MA, Habashi HO, Green AR, et al. Triple-negative breast cancer: distinguishing between basal and nonbasal subtypes. Clin Cancer Res. 2009;15:2302–2310. - PubMed
-
- Cheang MC, Voduc D, Bajdik C, Leung S, McKinney S, et al. Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res. 2008;14:1368–1376. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

