APPswe/Aβ regulation of osteoclast activation and RAGE expression in an age-dependent manner
- PMID: 21542009
- PMCID: PMC3126661
- DOI: 10.1002/jbmr.299
APPswe/Aβ regulation of osteoclast activation and RAGE expression in an age-dependent manner
Abstract
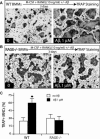
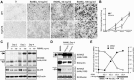
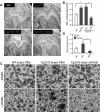
Alzheimer's disease (AD), one of the most dreaded neurodegenerative disorders, is characterized by cortical and cerebrovascular amyloid β peptide (Aβ) deposits, neurofibrillary tangles, chronic inflammation, and neuronal loss. Increased bone fracture rates and reduced bone density are commonly observed in patients with AD, suggesting one or more common denominators between both disorders. However, very few studies are available that have addressed this issue. Here, we present evidence for a function of amyloid precursor protein (APP) and Aβ in regulating osteoclast (OC) differentiation in vitro and in vivo. Tg2576 mice, which express the Swedish mutation of APP (APPswe) under the control of a prion promoter, exhibit biphasic effects on OC activation, with an increase of OCs in younger mice (< 4 months old), but a decrease in older Tg2576 mice (> 4 months old). The increase of OCs in young Tg2576 mice appears to be mediated by Aβ oligomers and receptor for advanced glycation end products (RAGE) expression in bone marrow macrophages (BMMs). However, the decrease of OC formation and activity in older Tg2576 mice may be due to the increase of soluble rage (sRAGE) in aged Tg2576 mice, an inhibitor of RANKL-induced osteoclastogenesis. These results suggest an unexpected function of APPswe/Aβ, reveal a mechanism underlying altered bone remodeling in AD patients, and implicate APP/Aβ and RAGE as common denominators for both AD and osteoporosis.
Copyright © 2011 American Society for Bone and Mineral Research.
Figures
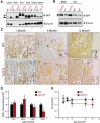
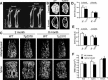
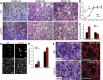

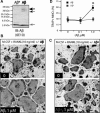



Similar articles
-
Amyloid β Peptide Enhances RANKL-Induced Osteoclast Activation through NF-κB, ERK, and Calcium Oscillation Signaling.Int J Mol Sci. 2016 Oct 10;17(10):1683. doi: 10.3390/ijms17101683. Int J Mol Sci. 2016. PMID: 27735865 Free PMC article.
-
Swedish mutant APP suppresses osteoblast differentiation and causes osteoporotic deficit, which are ameliorated by N-acetyl-L-cysteine.J Bone Miner Res. 2013 Oct;28(10):2122-35. doi: 10.1002/jbmr.1954. J Bone Miner Res. 2013. PMID: 23649480 Free PMC article.
-
DOK3 Modulates Bone Remodeling by Negatively Regulating Osteoclastogenesis and Positively Regulating Osteoblastogenesis.J Bone Miner Res. 2017 Nov;32(11):2207-2218. doi: 10.1002/jbmr.3205. Epub 2017 Aug 2. J Bone Miner Res. 2017. PMID: 28650106 Free PMC article.
-
Preventing activation of receptor for advanced glycation endproducts in Alzheimer's disease.Curr Drug Targets CNS Neurol Disord. 2005 Jun;4(3):249-66. doi: 10.2174/1568007054038210. Curr Drug Targets CNS Neurol Disord. 2005. PMID: 15975028 Review.
-
Ectodomain shedding of the receptor for advanced glycation end products: a novel therapeutic target for Alzheimer's disease.Cell Mol Life Sci. 2009 Dec;66(24):3923-35. doi: 10.1007/s00018-009-0121-4. Cell Mol Life Sci. 2009. PMID: 19672558 Free PMC article. Review.
Cited by
-
Amyloid β Peptide Enhances RANKL-Induced Osteoclast Activation through NF-κB, ERK, and Calcium Oscillation Signaling.Int J Mol Sci. 2016 Oct 10;17(10):1683. doi: 10.3390/ijms17101683. Int J Mol Sci. 2016. PMID: 27735865 Free PMC article.
-
Alzheimer's disease and osteoporosis.Tzu Chi Med J. 2017 Jul-Sep;29(3):138-142. doi: 10.4103/tcmj.tcmj_54_17. Tzu Chi Med J. 2017. PMID: 28974906 Free PMC article. Review.
-
Amyloid-β peptide treatment reverses bone loss in the mandibular condyle via Wnt signalling pathway.Bone Rep. 2024 Jul 8;22:101788. doi: 10.1016/j.bonr.2024.101788. eCollection 2024 Sep. Bone Rep. 2024. PMID: 39108841 Free PMC article.
-
Musculoskeletal Deficits and Cognitive Impairment: Epidemiological Evidence and Biological Mechanisms.Curr Osteoporos Rep. 2022 Oct;20(5):260-272. doi: 10.1007/s11914-022-00736-9. Epub 2022 Jun 29. Curr Osteoporos Rep. 2022. PMID: 35764750 Free PMC article. Review.
-
Autophagy alleviates the decrease in proliferation of amyloid β1‑42‑treated bone marrow mesenchymal stem cells via the AKT/mTOR signaling pathway.Mol Med Rep. 2019 May;19(5):4091-4100. doi: 10.3892/mmr.2019.10069. Epub 2019 Mar 21. Mol Med Rep. 2019. PMID: 30896831 Free PMC article.
References
-
- Rockenstein E, Crews L, Masliah E. Transgenic animal models of neurodegenerative diseases and their application to treatment development. Adv Drug Deliv Rev. 2007;59:1093–1102. - PubMed
-
- Hsiao KK, Borchelt DR, Olson K, et al. Age‐related CNS disorder and early death in transgenic FVB/N mice overexpressing Alzheimer amyloid precursor proteins. Neuron. 1995;15:1203–1218. - PubMed
-
- Tysiewicz‐Dudek M, Pietraszkiewicz F, Drozdzowska B. Alzheimer's disease and osteoporosis: common risk factors or one condition predisposing to the other? Ortop Traumatol Rehabil. 2008;10:315–323. - PubMed
-
- Melton LJ 3rd, Beard CM, Kokmen E, Atkinson EJ, O'Fallon WM. Fracture risk in patients with Alzheimer's disease. J Am Geriatr Soc. 1994;42:614–619. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

