HIV-1 Nef disrupts intracellular trafficking of major histocompatibility complex class I, CD4, CD8, and CD28 by distinct pathways that share common elements
- PMID: 21543478
- PMCID: PMC3126561
- DOI: 10.1128/JVI.00229-11
HIV-1 Nef disrupts intracellular trafficking of major histocompatibility complex class I, CD4, CD8, and CD28 by distinct pathways that share common elements
Abstract
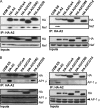
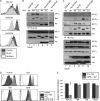
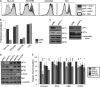
The Nef protein is an important HIV virulence factor that promotes the degradation of host proteins to augment virus production and facilitate immune evasion. The best-characterized targets of Nef are major histocompatibility complex class I (MHC-I) and CD4, but Nef also has been reported to target several other proteins, including CD8β, CD28, CD80, CD86, and CD1d. To compare and contrast the effects of Nef on each protein, we constructed a panel of chimeric proteins in which the extracellular and transmembrane regions of the MHC-I allele HLA-A2 were fused to the cytoplasmic tails of CD4, CD28, CD8β, CD80, CD86, and CD1d. We found that Nef coprecipitated with and disrupted the expression of molecules with cytoplasmic tails from MHC-I HLA-A2, CD4, CD8β, and CD28, but Nef did not bind to or alter the expression of molecules with cytoplasmic tails from CD80, CD86, and CD1d. In addition, we used short interfering RNA (siRNA) knockdown and coprecipitation experiments to implicate AP-1 as a cellular cofactor for Nef in the downmodulation of both CD28 and CD8β. The interaction with AP-1 required for CD28 and CD8β differed from the AP-1 interaction required for MHC-I downmodulation in that it was mediated through the dileucine motif within Nef (LL(164,165)AA) and did not require the tyrosine binding pocket of the AP-1 μ subunit. In addition, we demonstrate a requirement for β-COP as a cellular cofactor for Nef that was necessary for the degradation of targeted molecules HLA-A2, CD4, and CD8. These studies provide important new information on the similarities and differences with which Nef affects intracellular trafficking and help focus future research on the best potential pharmaceutical targets.
Figures





References
-
- Arold S. T., Baur A. S. 2001. Dynamic Nef and Nef dynamics: how structure could explain the complex activities of this small HIV protein. Trends Biochem. Sci. 26:356–363 - PubMed
-
- Benichou S., et al. 1994. Physical interaction of the HIV-1 Nef protein wih β-cop, a component of non-clathrin coated vesicles essential for membrane traffic. J. Biol. Chem. 269:30073–30076 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

