Immune and Genetic Correlates of Vaccine Protection Against Mucosal Infection by SIV in Monkeys
- PMID: 21543722
- PMCID: PMC3718279
- DOI: 10.1126/scitranslmed.3002351
Immune and Genetic Correlates of Vaccine Protection Against Mucosal Infection by SIV in Monkeys
Abstract
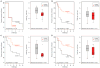
The RV144 vaccine trial in Thailand demonstrated that an HIV vaccine could prevent infection in humans and highlights the importance of understanding protective immunity against HIV. We used a nonhuman primate model to define immune and genetic mechanisms of protection against mucosal infection by the simian immunodeficiency virus (SIV). A plasmid DNA prime/recombinant adenovirus serotype 5 (rAd5) boost vaccine regimen was evaluated for its ability to protect monkeys from infection by SIVmac251 or SIVsmE660 isolates after repeat intrarectal challenges. Although this prime-boost vaccine regimen failed to protect against SIVmac251 infection, 50% of vaccinated monkeys were protected from infection with SIVsmE660. Among SIVsmE660-infected animals, there was about a one-log reduction in peak plasma virus RNA in monkeys expressing the major histocompatibility complex class I allele Mamu-A*01, implicating cytotoxic T lymphocytes in the control of SIV replication once infection is established. Among Mamu-A*01-negative monkeys challenged with SIVsmE660, no CD8(+) T cell response or innate immune response was associated with protection against virus acquisition. However, low levels of neutralizing antibodies and an envelope-specific CD4(+) T cell response were associated with vaccine protection in these monkeys. Moreover, monkeys that expressed two TRIM5 alleles that restrict SIV replication were more likely to be protected from infection than monkeys that expressed at least one permissive TRIM5 allele. This study begins to elucidate the mechanisms of vaccine protection against immunodeficiency viruses and highlights the need to analyze these immune and genetic correlates of protection in future trials of HIV vaccine strategies.
Conflict of interest statement
Figures


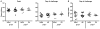





References
-
- Flynn NM, Forthal DN, Harro CD, Judson FN, Mayer KH, Para MF. rgp120 HIV Vaccine Study Group, Placebo-controlled phase 3 trial of a recombinant glycoprotein 120 vaccine to prevent HIV-1 infection. J Infect Dis. 2005;191:654–665. - PubMed
-
- Pitisuttithum P, Gilbert P, Gurwith M, Heyward W, Martin M, van Griensven F, Hu D, Tappero JW, Choopanya K. Bangkok Vaccine Evaluation Group, Randomized, double-blind, placebo-controlled efficacy trial of a bivalent recombinant glycoprotein 120 HIV-1 vaccine among injection drug users in Bangkok, Thailand. J Infect Dis. 2006;194:1661–1671. - PubMed
-
- Buchbinder SP, Mehrotra DV, Duerr A, Fitzgerald DW, Mogg R, Li D, Gilbert PB, Lama JR, Marmor M, Del Rio C, McElrath MJ, Casimiro DR, Gottesdiener KM, Chodakewitz JA, Corey L, Robertson MN. Step Study Protocol Team, Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): A double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet. 2008;372:1881–1893. - PMC - PubMed
-
- McElrath MJ, De Rosa SC, Moodie Z, Dubey S, Kierstead L, Janes H, Defawe OD, Carter DK, Hural J, Akondy R, Buchbinder SP, Robertson MN, Mehrotra DV, Self SG, Corey L, Shiver JW, Casimiro DR. Step Study Protocol Team, HIV-1 vaccine-induced immunity in the test-of-concept Step Study: A case-cohort analysis. Lancet. 2008;372:1894–1905. - PMC - PubMed
-
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E, Benenson M, Gurunathan S, Tartaglia J, McNeil JG, Francis DP, Stablein D, Birx DL, Chunsuttiwat S, Khamboonruang C, Thongcharoen P, Robb ML, Michael NL, Kunasol P, Kim JH. MOPH-TAVEG Investigators, Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N Engl J Med. 2009;361:2209–2220. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

