Proteostasis: a new therapeutic paradigm for pulmonary disease
- PMID: 21543800
- PMCID: PMC3131838
- DOI: 10.1513/pats.201008-055MS
Proteostasis: a new therapeutic paradigm for pulmonary disease
Abstract
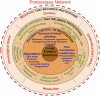
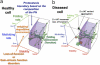
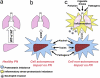
Among lung pathologies, α1AT, chronic obstructive pulmonary disease (COPD), emphysema, and asthma are diseases triggered by local environmental stress in the airway that we refer to herein collectively as airway stress diseases (ASDs). A deficiency of α-1-antitrypsin (α1AT) is an inherited genetic disorder that is a consequence of the misfolding of α1AT during protein synthesis in liver hepatocytes, reducing secretion to the plasma and delivery to the lung. Deficiency of α1AT in the lung triggers a similar pathological phenotype to other ASDs. Moreover, the loss of α1AT in the lung is a well-known environmental risk factor for COPD/emphysema. To date there are no effective therapeutic approaches to address ASDs, which reflects a general lack of understanding of their cellular basis. Herein, we propose that ASDs are disorders of proteostasis. That is, they are initiated and propagated by a common theme-a challenge to protein folding capacity maintained by the proteostasis network (PN) (see Balch et al., Science 2008;319:916-919). The PN is a network of chaperones and degradative components that generates and manages protein folding pathways responsible for normal human physiology. In ASD, we suggest that the PN system fails to respond to the increased burden of unfolded proteins due to genetic and environmental stresses, thus triggering pulmonary pathophysiology. We introduce the enabling concept of proteostasis regulators (PRs), small molecules that regulate signaling pathways that control the composition and activity of PN components, as a new and general approach for therapeutic management of ASDs.
Figures
References
Publication types
MeSH terms
Grants and funding
- HL079442/HL/NHLBI NIH HHS/United States
- R01 GM042336/GM/NIGMS NIH HHS/United States
- AG18917/AG/NIA NIH HHS/United States
- P01 AG031097/AG/NIA NIH HHS/United States
- AG036634/AG/NIA NIH HHS/United States
- DK051870/DK/NIDDK NIH HHS/United States
- R01 HL095524/HL/NHLBI NIH HHS/United States
- R01 GM033301/GM/NIGMS NIH HHS/United States
- HL095524/HL/NHLBI NIH HHS/United States
- GM42336/GM/NIGMS NIH HHS/United States
- R21 NS067643/NS/NINDS NIH HHS/United States
- AG031097/AG/NIA NIH HHS/United States
- RC2 AG036634/AG/NIA NIH HHS/United States
- NS067643/NS/NINDS NIH HHS/United States
- R01 HL079442/HL/NHLBI NIH HHS/United States
- GM33301/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
