The adenovirus E4orf4 protein targets PP2A to the ACF chromatin-remodeling factor and induces cell death through regulation of SNF2h-containing complexes
- PMID: 21546548
- PMCID: PMC3159439
- DOI: 10.1093/nar/gkr231
The adenovirus E4orf4 protein targets PP2A to the ACF chromatin-remodeling factor and induces cell death through regulation of SNF2h-containing complexes
Abstract
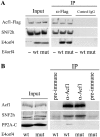
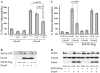
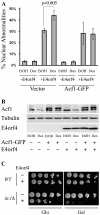
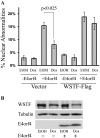
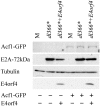
The adenovirus E4 open-reading-frame 4 (E4orf4) protein regulates the progression of viral infection and when expressed individually it induces non-classical apoptosis in transformed cells. Here we show that E4orf4 associates with the ATP-dependent chromatin-remodeling factor ACF that consists of a sucrose non fermenting-2h (SNF2h) ATPase and an Acf1 regulatory subunit. Furthermore, E4orf4 targets protein phosphatase 2A (PP2A) to this complex and to chromatin. Obstruction of SNF2h activity inhibits E4orf4-induced cell death, whereas knockdown of Acf1 results in enhanced E4orf4-induced toxicity in both mammalian and yeast cells, and Acf1 overexpression inhibits E4orf4's ability to downregulate early adenovirus gene expression in the context of viral infection. Knockdown of the Acf1 homolog, WSTF, inhibits E4orf4-induced cell death. Based on these results we suggest that the E4orf4-PP2A complex inhibits ACF and facilitates enhanced chromatin-remodeling activities of other SNF2h-containing complexes, such as WSTF-SNF2h. The resulting switch in chromatin remodeling determines life versus death decisions and contributes to E4orf4 functions during adenovirus infection.
Figures

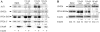


References
-
- Mannervik M, Fan S, Strom AC, Helin K, Akusjarvi G. Adenovirus E4 open reading frame 4-induced dephosphorylation inhibits E1A activation of the E2 promoter and E2F-1-mediated transactivation independently of the retinoblastoma tumor suppressor protein. Virology. 1999;256:313–321. - PubMed
-
- Kanopka A, Muhlemann O, Petersen-Mahrt S, Estmer C, Ohrmalm C, Akusjarvi G. Nature. 1998;393:185–187. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

