Nerve-derived sonic hedgehog defines a niche for hair follicle stem cells capable of becoming epidermal stem cells
- PMID: 21549329
- PMCID: PMC3089905
- DOI: 10.1016/j.stem.2011.02.021
Nerve-derived sonic hedgehog defines a niche for hair follicle stem cells capable of becoming epidermal stem cells
Abstract
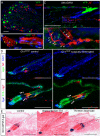
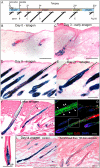
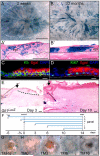
In adult skin, stem cells in the hair follicle bulge cyclically regenerate the follicle, whereas a distinct stem cell population maintains the epidermis. The degree to which all bulge cells have equal regenerative potential is not known. We found that Sonic hedgehog (Shh) from neurons signals to a population of cells in the telogen bulge marked by the Hedgehog response gene Gli1. Gli1-expressing bulge cells function as multipotent stem cells in their native environment and repeatedly regenerate the anagen follicle. Shh-responding perineural bulge cells incorporate into healing skin wounds where, notably, they can change their lineage into epidermal stem cells. The perineural niche (including Shh) is dispensable for follicle contributions to acute wound healing and skin homeostasis, but is necessary to maintain bulge cells capable of becoming epidermal stem cells. Thus, nerves cultivate a microenvironment where Shh creates a molecularly and phenotypically distinct population of hair follicle stem cells.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures




Comment in
-
A nervous hedgehog rolls into the hair follicle stem cell scene.Cell Stem Cell. 2011 May 6;8(5):459-60. doi: 10.1016/j.stem.2011.04.005. Cell Stem Cell. 2011. PMID: 21549317 No abstract available.
References
-
- Adolphe C, Narang M, Ellis T, Wicking C, Kaur P, Wainwright B. An in vivo comparative study of sonic, desert and Indian hedgehog reveals that hedgehog pathway activity regulates epidermal stem cell homeostasis. Development. 2004;131:5009–5019. - PubMed
-
- Ahn S, Joyner AL. Dynamic changes in the response of cells to positive hedgehog signaling during mouse limb patterning. Cell. 2004;118:505–516. - PubMed
-
- Ahn S, Joyner AL. In vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nature. 2005;437:894–897. - PubMed
-
- Bai CB, Auerbach W, Lee JS, Stephen D, Joyner AL. Gli2, but not Gli1, is required for initial Shh signaling and ectopic activation of the Shh pathway. Development. 2002;129:4753–4761. - PubMed
-
- Bai CB, Joyner AL. Gli1 can rescue the in vivo function of Gli2. Development. 2001;128:5161–5172. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

