Exome sequencing and functional analysis identifies BANF1 mutation as the cause of a hereditary progeroid syndrome
- PMID: 21549337
- PMCID: PMC3146734
- DOI: 10.1016/j.ajhg.2011.04.010
Exome sequencing and functional analysis identifies BANF1 mutation as the cause of a hereditary progeroid syndrome
Abstract
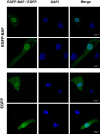
Accelerated aging syndromes represent a valuable source of information about the molecular mechanisms involved in normal aging. Here, we describe a progeroid syndrome that partially phenocopies Hutchinson-Gilford progeria syndrome (HGPS) but also exhibits distinctive features, including the absence of cardiovascular deficiencies characteristic of HGPS, the lack of mutations in LMNA and ZMPSTE24, and a relatively long lifespan of affected individuals. Exome sequencing and molecular analysis in two unrelated families allowed us to identify a homozygous mutation in BANF1 (c.34G>A [p.Ala12Thr]), encoding barrier-to-autointegration factor 1 (BAF), as the molecular abnormality responsible for this Mendelian disorder. Functional analysis showed that fibroblasts from both patients have a dramatic reduction in BAF protein levels, indicating that the p.Ala12Thr mutation impairs protein stability. Furthermore, progeroid fibroblasts display profound abnormalities in the nuclear lamina, including blebs and abnormal distribution of emerin, an interaction partner of BAF. These nuclear abnormalities are rescued by ectopic expression of wild-type BANF1, providing evidence for the causal role of this mutation. These data demonstrate the utility of exome sequencing for identifying the cause of rare Mendelian disorders and underscore the importance of nuclear envelope alterations in human aging.
Copyright © 2011 The American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Martin G.M., Oshima J. Lessons from human progeroid syndromes. Nature. 2000;408:263–266. - PubMed
-
- Burtner C.R., Kennedy B.K. Progeria syndromes and ageing: what is the connection? Nat. Rev. Mol. Cell Biol. 2010;11:567–578. - PubMed
-
- De Sandre-Giovannoli A., Bernard R., Cau P., Navarro C., Amiel J., Boccaccio I., Lyonnet S., Stewart C.L., Munnich A., Le Merrer M., Lévy N. Lamin a truncation in Hutchinson-Gilford progeria. Science. 2003;300:2055. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

