Release mode of large and small dense-core vesicles specified by different synaptotagmin isoforms in PC12 cells
- PMID: 21551071
- PMCID: PMC3128534
- DOI: 10.1091/mbc.E11-02-0159
Release mode of large and small dense-core vesicles specified by different synaptotagmin isoforms in PC12 cells
Abstract
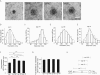
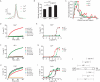
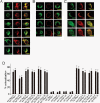
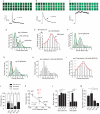
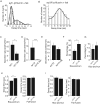
Many cells release multiple substances in different proportions according to the specific character of a stimulus. PC12 cells, a model neuroendocrine cell line, express multiple isoforms of the exocytotic Ca(2+) sensor synaptotagmin. We show that these isoforms sort to populations of dense-core vesicles that differ in size. These synaptotagmins differ in their Ca(2+) sensitivities, their preference for full fusion or kiss-and-run, and their sensitivity to inhibition by synaptotagmin IV. In PC12 cells, vesicles that harbor these different synaptotagmin isoforms can be preferentially triggered to fuse by different forms of stimulation. The mode of fusion is specified by the synaptotagmin isoform activated, and because kiss-and-run exocytosis can filter small molecules through a size-limiting fusion pore, the activation of isoforms that favor kiss-and-run will select smaller molecules over larger molecules packaged in the same vesicle. Thus synaptotagmin isoforms can provide multiple levels of control in the release of different molecules from the same cell.
Figures


References
-
- Alvarez de Toledo G, Fernandez-Chacon R, Fernandez JM. Release of secretory products during transient vesicle fusion. Nature. 1993;363:554–558. - PubMed
-
- Ariga T, Macala LJ, Saito M, Margolis RK, Greene LA, Margolis RU, Yu RK. Lipid composition of PC12 pheochromocytoma cells: characterization of globoside as a major neutral glycolipid. Biochemistry. 1988;27:52–58. - PubMed
-
- Augustine GJ. How does calcium trigger neurotransmitter release? Curr Opin Neurobiol. 2001;11:320–326. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

