Eukaryotic-like Ser/Thr protein kinases SpkC/F/K are involved in phosphorylation of GroES in the Cyanobacterium synechocystis
- PMID: 21551175
- PMCID: PMC3111230
- DOI: 10.1093/dnares/dsr006
Eukaryotic-like Ser/Thr protein kinases SpkC/F/K are involved in phosphorylation of GroES in the Cyanobacterium synechocystis
Abstract
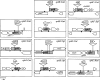
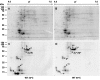
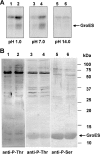
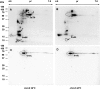
Serine/threonine protein kinases (STPKs) are the major participants in intracellular signal transduction in eukaryotes, such as yeasts, fungi, plants, and animals. Genome sequences indicate that these kinases are also present in prokaryotes, such as cyanobacteria. However, their roles in signal transduction in prokaryotes remain poorly understood. We have attempted to identify the roles of STPKs in response to heat stress in the prokaryotic cyanobacterium Synechocystis sp. PCC 6803, which has 12 genes for STPKs. Each gene was individually inactivated to generate a gene-knockout library of STPKs. We applied in vitro Ser/Thr protein phosphorylation and phosphoproteomics and identified the methionyl-tRNA synthetase, large subunit of RuBisCO, 6-phosphogluconate dehydrogenase, translation elongation factor Tu, heat-shock protein GrpE, and small chaperonin GroES as the putative targets for Ser/Thr phosphorylation. The expressed and purified GroES was used as an external substrate to screen the protein extracts of the individual mutants for their Ser/Thr kinase activities. The mutants that lack one of the three protein kinases, SpkC, SpkF, and SpkK, were unable to phosphorylate GroES in vitro, suggesting possible interactions between them towards their substrate. Complementation of the mutated SpkC, SpkF, and SpkK leads to the restoration of the ability of cells to phosphorylate the GroES. This suggests that these three STPKs are organized in a sequential order or a cascade and they work one after another to finally phosphorylate the GroES.
Figures


Similar articles
-
Post-translational Modifications of Serine/Threonine and Histidine Kinases and Their Roles in Signal Transductions in Synechocystis Sp. PCC 6803.Appl Biochem Biotechnol. 2021 Mar;193(3):687-716. doi: 10.1007/s12010-020-03435-2. Epub 2020 Nov 6. Appl Biochem Biotechnol. 2021. PMID: 33159456 Review.
-
Biochemical examination of the potential eukaryotic-type protein kinase genes in the complete genome of the unicellular Cyanobacterium synechocystis sp. PCC 6803.DNA Res. 2002 Jun 30;9(3):71-8. doi: 10.1093/dnares/9.3.71. DNA Res. 2002. PMID: 12168951
-
Genomic analysis of protein kinases, protein phosphatases and two-component regulatory systems of the cyanobacterium Anabaena sp. strain PCC 7120.FEMS Microbiol Lett. 2002 Dec 17;217(2):155-65. doi: 10.1111/j.1574-6968.2002.tb11469.x. FEMS Microbiol Lett. 2002. PMID: 12480098
-
On the origin of Ser/Thr kinases in a prokaryote.FEMS Microbiol Lett. 2001 Jun 12;200(1):79-84. doi: 10.1111/j.1574-6968.2001.tb10696.x. FEMS Microbiol Lett. 2001. PMID: 11410353
-
Signaling mechanisms of the Mycobacterium tuberculosis receptor Ser/Thr protein kinases.Curr Opin Struct Biol. 2009 Dec;19(6):650-7. doi: 10.1016/j.sbi.2009.10.017. Epub 2009 Nov 14. Curr Opin Struct Biol. 2009. PMID: 19914822 Free PMC article. Review.
Cited by
-
The Cytochrome b 6 f Complex Is Not Involved in Cyanobacterial State Transitions.Plant Cell. 2019 Apr;31(4):911-931. doi: 10.1105/tpc.18.00916. Epub 2019 Mar 8. Plant Cell. 2019. PMID: 30852554 Free PMC article.
-
Thiol-based redox modulation of a cyanobacterial eukaryotic-type serine/threonine kinase required for oxidative stress tolerance.Antioxid Redox Signal. 2012 Aug 15;17(4):521-33. doi: 10.1089/ars.2011.4483. Epub 2012 Jun 4. Antioxid Redox Signal. 2012. PMID: 22530622 Free PMC article.
-
Revisiting cyanobacterial state transitions.Photochem Photobiol Sci. 2020 May 1;19(5):585-603. doi: 10.1039/c9pp00451c. Epub 2020 Mar 12. Photochem Photobiol Sci. 2020. PMID: 32163064 Review.
-
6-Phosphogluconate dehydrogenase and its crystal structures.Acta Crystallogr F Struct Biol Commun. 2022 Mar 1;78(Pt 3):96-112. doi: 10.1107/S2053230X22001091. Epub 2022 Feb 23. Acta Crystallogr F Struct Biol Commun. 2022. PMID: 35234135 Free PMC article. Review.
-
Distinct Molecular Patterns of Two-Component Signal Transduction Systems in Thermophilic Cyanobacteria as Revealed by Genomic Identification.Biology (Basel). 2023 Feb 8;12(2):271. doi: 10.3390/biology12020271. Biology (Basel). 2023. PMID: 36829548 Free PMC article.
References
-
- Dutta R., Quin L., Inouye M. Histidine kinases: diversity of domain organization. Mol. Microbiol. 1999;34:633–40. doi:10.1046/j.1365-2958.1999.01646.x. - DOI - PubMed
-
- Los D.A., Zorina A., Sinetova M., Kryazhov S., Mironov K., Zinchenko V.V. Stress sensors and signal transducers in cyanobacteria. Sensors. 2010;10:2386–415. doi:10.3390/s100302386. - DOI - PMC - PubMed
-
- Hanks S.K., Hunter T. The eukaryotic protein kinase superfamily: kinase (catalytic) domain structure and classification. FASEB J. 1995;9:576–96. - PubMed
-
- Kennelly P.J. Protein kinases and protein phosphatases in prokaryotes: a genomic perspective. FEMS Microbiol. Lett. 2002;206:1–8. doi:10.1111/j.1574-6968.2002.tb10978.x. - DOI - PubMed
-
- Krupa A., Abhinandan K.R., Srinivasan N. KinG: a database of protein kinases in genomes. Nucleic Acids Res. 2004;32:D153–5. http://hodgkin.mbu.iisc.ernet.in/king2/cgi-bin/index. doi:10.1093/nar/gkh019. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

