Perinatal or adult Nf1 inactivation using tamoxifen-inducible PlpCre each cause neurofibroma formation
- PMID: 21551249
- PMCID: PMC3464476
- DOI: 10.1158/0008-5472.CAN-10-4558
Perinatal or adult Nf1 inactivation using tamoxifen-inducible PlpCre each cause neurofibroma formation
Abstract
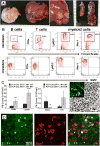
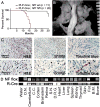
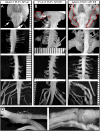
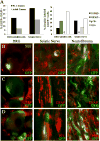
Plexiform neurofibromas are peripheral nerve sheath tumors initiated by biallelic mutation of the NF1 tumor suppressor gene in the Schwann cell lineage. To understand whether neurofibroma formation is possible after birth, we induced Nf1 loss of function with an inducible proteolipid protein Cre allele. Perinatal loss of Nf1 resulted in the development of small plexiform neurofibromas late in life, whereas loss in adulthood caused large plexiform neurofibromas and morbidity beginning 4 months after onset of Nf1 loss. A conditional EGFP reporter allele identified cells showing recombination, including peripheral ganglia satellite cells, peripheral nerve S100β+ myelinating Schwann cells, and peripheral nerve p75+ cells. Neurofibromas contained cells with Remak bundle disruption but no recombination within GFAP+ nonmyelinating Schwann cells. Extramedullary lympho-hematopoietic expansion was also observed in PlpCre;Nf1fl/fl mice. These tumors contained EGFP+/Sca-1+ stromal cells among EGFP-negative lympho-hematopoietic cells indicating a noncell autonomous effect and unveiling a role of Nf1-deleted microenvironment on lympho-hematopoietic proliferation in vivo. Together these findings define a tumor suppressor role for Nf1 in the adult and narrow the range of potential neurofibroma-initiating cell populations.
©2011 AACR.
Figures


References
-
- Friedman JM. Epidemiology of neurofibromatosis type 1. Am J Med Genet. 1999;891:1–6. - PubMed
-
- Rosenfeld A, Listernick R, Charrow J, Goldman S. Neurofibromatosis type 1 and high-grade tumors of the central nervous system. Childs Nerv Syst. 2010;265:663–7. - PubMed
-
- Emanuel PD, Bates LJ, Castleberry RP, et al. Selective hypersensitivity to granulocyte-macrophage colony-stimulating factor by juvenile chronic myeloid leukemia hematopoietic progenitors. Blood. 1991;77:925–929. - PubMed
-
- Niemeyer CM, Arico M, Basso G, et al. Quantitative effects of Nf1 inactivation on in vivo hematopoiesis. Blood. 1997;89:3534.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

