Androgen Receptor regulation of Vitamin D receptor in response of castration-resistant prostate cancer cells to 1α-Hydroxyvitamin D5 - a calcitriol analog
- PMID: 21552398
- PMCID: PMC3089062
- DOI: 10.1177/1947601910385450
Androgen Receptor regulation of Vitamin D receptor in response of castration-resistant prostate cancer cells to 1α-Hydroxyvitamin D5 - a calcitriol analog
Abstract
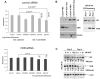
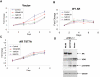
Calcitriol (1,25(OH)(2)D3) is cytostatic for prostate cancer (CaP), but had limited therapeutic utility due to hypercalcemia-related toxicities, leading to the development of low-calcemic calcitriol analogs. We show that one analog, 1-α-Hydroxyvitamin-D5 (1α(OH)D5), induced apoptosis in castration-sensitive LNCaP prostate cancer cells, but unlike calcitriol, did not increase androgen receptor (AR) transcriptional activity. LNCaP-AI, a castrate-resistant (CRCaP) LNCaP subline, was resistant to 1α(OH)D5 in the presence of androgens; however, androgen withdrawal (AWD), although ineffective by itself, sensitized LNCaP-AI cells to 1α(OH)D5. Investigation of the mechanism revealed that the vitamin D receptor (VDR), which mediates the effects of 1α(OH)D5, is downregulated in LNCaP-AI cells compared to LNCaP in the presence of androgens, whereas AWD restored VDR expression. Since LNCaP-AI cells expressed higher AR compared to LNCaP and AWD decreased AR, this indicated an inverse relationship between VDR and AR. Further, AR stimulation (by increased androgen) suppressed VDR, while AR downregulation (by ARsiRNA) stimulated VDR levels and sensitized LNCaP-AI cells to 1α(OH)D5 similar to AWD. Another cell line, pRNS-1-1, although isolated from a normal prostate, had lost AR expression in culture and adapted to androgen-independent growth. These cells expressed the VDR and were sensitive to 1α(OH)D5, but restoration of AR expression suppressed VDR levels and induced resistance to 1α(OH)D5 treatment. Taken together, these results demonstrate negative regulation of VDR by AR in CRCaP cells. This effect is likely mediated by prohibitin (PHB), which was inhibited by AR transcriptional activity and stimulated VDR in CRCaP, but not castrate-sensitive cells. Therefore, in castration sensitive cells, although the AR negatively regulates PHB, this does not affect VDR expression, whereas in CRCaP cells, negative regulation of PHB by the AR results in concomitant negative regulation of the VDR by the AR. These data demonstrate a novel mechanism by which 1α(OH)D5 prolong the effectiveness of AWD in CaP cells.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Figures




References
-
- Edwards J, Bartlett JM. The androgen receptor and signal-transduction pathways in hormone-refractory prostate cancer. Part 1: modifications to the androgen receptor. BJU Int. 2005;95:1320-6 - PubMed
-
- Klotz L. Maximal androgen blockade for advanced prostate cancer. Best Pract Res Clin Endocrinol Metab. 2008;22:331-40 - PubMed
-
- Feldman BJ, Feldman D. The development of androgen-independent prostate cancer. Nat Rev Cancer. 2001;1:34-45 - PubMed
-
- Petrylak D. Therapeutic options in androgen-independent prostate cancer: building on docetaxel. BJU Int. 2005;96 Suppl 2:41-6 - PubMed
-
- Boehmer A, Anastasiadis AG, Feyerabend S, et al. Docetaxel, estramustine and prednisone for hormone-refractory prostate cancer: a single-center experience. Anticancer Res. 2005;25:4481-6 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
