Wildtype and engineered monomeric triosephosphate isomerase from Trypanosoma brucei: partitioning of reaction intermediates in D2O and activation by phosphite dianion
- PMID: 21553855
- PMCID: PMC3138527
- DOI: 10.1021/bi2005416
Wildtype and engineered monomeric triosephosphate isomerase from Trypanosoma brucei: partitioning of reaction intermediates in D2O and activation by phosphite dianion
Abstract
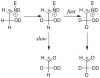
Product yields for the reactions of (R)-glyceraldehyde 3-phosphate (GAP) in D2O at pD 7.9 catalyzed by wildtype triosephosphate isomerase from Trypanosoma brucei brucei (Tbb TIM) and a monomeric variant (monoTIM) of this wildtype enzyme were determined by (1)H NMR spectroscopy and were compared with the yields determined in earlier work for the reactions catalyzed by TIM from rabbit and chicken muscle [O'Donoghue, A. C., Amyes, T. L., and Richard, J. P. (2005), Biochemistry 44, 2610 - 2621]. Three products were observed from the reactions catalyzed by TIM: dihydroxyacetone phosphate (DHAP) from isomerization with intramolecular transfer of hydrogen, d-DHAP from isomerization with incorporation of deuterium from D2O into C-1 of DHAP, and d-GAP from incorporation of deuterium from D2O into C-2 of GAP. The yield of DHAP formed by intramolecular transfer of hydrogen decreases from 49% for the muscle enzymes to 40% for wildtype Tbb TIM to 34% for monoTIM. There is no significant difference in the ratio of the yields of d-DHAP and d-GAP for wildtype TIM from muscle sources and Trypanosoma brucei brucei, but partitioning of the enediolate intermediate of the monoTIM reaction to form d-DHAP is less favorable ((k(C1))(D)/(k(C2))(D) = 1.1) than for the wildtype enzyme ((k(C1))(D)/(k(C2))(D) = 1.7). Product yields for the wildtype Tbb TIM and monoTIM-catalyzed reactions of glycolaldehyde labeled with carbon-13 at the carbonyl carbon ([1-(13)C]-GA) at pD 7.0 in the presence of phosphite dianion and in its absence were determined by (1)H NMR spectroscopy [Go, M. K., Amyes, T. L., and Richard, J. P. (2009) Biochemistry 48, 5769-5778]. There is no detectable difference in the yields of the products of wildtype muscle and Tbb TIM-catalyzed reactions of [1-(13)C]-GA in D2O. The kinetic parameters for phosphite dianion activation of the reactions of [1-(13)C]-GA catalyzed by wildtype Tbb TIM are similar to those reported for the enzyme from rabbit muscle [Amyes, T. L. and Richard, J. P. (2007) Biochemistry 46, 5841-5854], but there is no detectable dianion activation of the reaction catalyzed by monoTIM. The engineered disruption of subunit contacts at monoTIM causes movement of the essential side chains of Lys-13 and His-95 away from the catalytic active positions. We suggest that this places an increased demand that the intrinsic binding energy of phosphite dianion be utilized to drive the change in the conformation of monoTIM back to the active structure for wildtype TIM.
Figures










References
-
- Knowles JR, Albery WJ. Perfection in enzyme catalysis: the energetics of triosephosphate isomerase. Acc Chem Res. 1977;10:105–111.
-
- Rieder SV, Rose IA. Mechanism of the triose phosphate isomerase reaction. J Biol Chem. 1959;234:1007–1010. - PubMed
-
- Amyes TL, O’Donoghue AC, Richard JP. Contribution of phosphate intrinsic binding energy to the enzymatic rate acceleration for triosephosphate isomerase. J Am Chem Soc. 2001;123:11325–11326. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

