Wiring and rewiring of the retinogeniculate synapse
- PMID: 21558027
- PMCID: PMC3099477
- DOI: 10.1016/j.conb.2011.02.007
Wiring and rewiring of the retinogeniculate synapse
Abstract
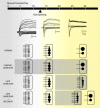
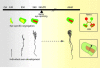
The formation and refinement of synaptic circuits are areas of research that have fascinated neurobiologists for decades. A recurrent theme seen at many CNS synapses is that neuronal connections are at first imprecise, but refine and can be rearranged with time or with experience. Today, with the advent of new technologies to map and monitor neuronal circuits, it is worthwhile to revisit a powerful experimental model for examining the development and plasticity of synaptic circuits--the retinogeniculate synapse.
Copyright © 2011 Elsevier Ltd. All rights reserved.
Figures


References
-
- Kano M, Hashimoto K. Synapse elimination in the central nervous system. Curr Opin Neurobiol. 2009;19:154–161. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

