Mechanism of a genetically encoded dark-to-bright reporter for caspase activity
- PMID: 21558267
- PMCID: PMC3137071
- DOI: 10.1074/jbc.M111.221648
Mechanism of a genetically encoded dark-to-bright reporter for caspase activity
Abstract
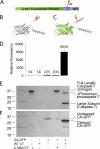
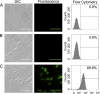
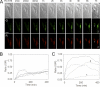
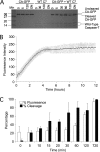
Fluorescent proteins have revolutionized modern biology with their ability to report the presence of tagged proteins in living systems. Although several fluorescent proteins have been described in which the excitation and emission properties can be modulated by external triggers, no fluorescent proteins have been described that can be activated from a silent dark state to a bright fluorescent state directly by the activity of an enzyme. We have developed a version of GFP in which fluorescence is completely quenched by appendage of a hydrophobic quenching peptide that tetramerizes GFP and prevents maturation of the chromophore. The fluorescence can be fully restored by catalytic removal of the quenching peptide, making it a robust reporter of proteolysis. We have demonstrated the utility of this uniquely dark state of GFP as a genetically encoded apoptosis reporter that monitors the function of caspases, which catalyze the fate-determining step in programmed cell death. Caspase Activatable-GFP (CA-GFP) can be activated both in vitro and in vivo, resulting in up to a 45-fold increase in fluorescent signal in bacteria and a 3-fold increase in mammalian cells. We used CA-GFP successfully to monitor real-time apoptosis in mammalian cells. This dark state of GFP may ultimately serve as a useful platform for probes of other enzymatic processes.
Figures


References
-
- Shaner N. C., Steinbach P. A., Tsien R. Y. (2005) Nat. Methods 2, 905–909 - PubMed
-
- Lukyanov K. A., Chudakov D. M., Lukyanov S., Verkhusha V. V. (2005) Nat. Reviews 6, 885–891 - PubMed
-
- Reed J. C., Tomaselli K. J. (2000) Curr Opin Biotechnol 11, 586–592 - PubMed
-
- Garcia-Calvo M., Peterson E. P., Rasper D. M., Vaillancourt J. P., Zamboni R., Nicholson D. W., Thornberry N. A. (1999) Cell Death Differ. 6, 362–369 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

