Evidence for a stromal-epithelial "lactate shuttle" in human tumors: MCT4 is a marker of oxidative stress in cancer-associated fibroblasts
- PMID: 21558814
- PMCID: PMC3142461
- DOI: 10.4161/cc.10.11.15659
Evidence for a stromal-epithelial "lactate shuttle" in human tumors: MCT4 is a marker of oxidative stress in cancer-associated fibroblasts
Abstract
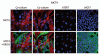
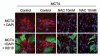
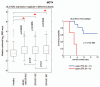
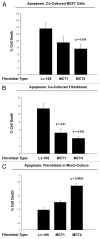
Recently, we proposed a new mechanism for understanding the Warburg effect in cancer metabolism. In this new paradigm, cancer-associated fibroblasts undergo aerobic glycolysis, and extrude lactate to "feed" adjacent cancer cells, which then drives mitochondrial biogenesis and oxidative mitochondrial metabolism in cancer cells. Thus, there is vectorial transport of energy-rich substrates from the fibroblastic tumor stroma to anabolic cancer cells. A prediction of this hypothesis is that cancer-associated fibroblasts should express MCT4, a mono-carboxylate transporter that has been implicated in lactate efflux from glycolytic muscle fibers and astrocytes in the brain. To address this issue, we co-cultured MCF7 breast cancer cells with normal fibroblasts. Interestingly, our results directly show that breast cancer cells specifically induce the expression of MCT4 in cancer-associated fibroblasts; MCF7 cells alone and fibroblasts alone, both failed to express MCT4. We also show that the expression of MCT4 in cancer-associated fibroblasts is due to oxidative stress, and can be prevented by pre-treatment with the anti-oxidant N-acetyl-cysteine. In contrast to our results with MCT4, we see that MCT1, a transporter involved in lactate uptake, is specifically upregulated in MCF7 breast cancer cells when co-cultured with fibroblasts. Virtually identical results were also obtained with primary human breast cancer samples. In human breast cancers, MCT4 selectively labels the tumor stroma, e.g., the cancer-associated fibroblast compartment. Conversely, MCT1 was selectively expressed in the epithelial cancer cells within the same tumors. Functionally, we show that overexpression of MCT4 in fibroblasts protects both MCF7 cancer cells and fibroblasts against cell death, under co-culture conditions. Thus, we provide the first evidence for the existence of a stromal-epithelial lactate shuttle in human tumors, analogous to the lactate shuttles that are essential for the normal physiological function of muscle tissue and brain. These data are consistent with the "reverse Warburg effect," which states that cancer-associated fibroblasts undergo aerobic glycolysis, thereby producing lactate, which is utilized as a metabolic substrate by adjacent cancer cells. In this model, "energy transfer" or "metabolic-coupling" between the tumor stroma and epithelial cancer cells "fuels" tumor growth and metastasis, via oxidative mitochondrial metabolism in anabolic cancer cells. Most importantly, our current findings provide a new rationale and novel strategy for anti-cancer therapies, by employing MCT inhibitors.
Figures






Comment in
-
Fuel for cancer cells?Cell Cycle. 2011 Aug 1;10(15):2421-2. doi: 10.4161/cc.10.15.16204. Epub 2011 Aug 1. Cell Cycle. 2011. PMID: 21734458 No abstract available.
References
-
- Gladden LB. A lactatic perspective on metabolism. Med Sci Sports Exerc. 2008;40:477–485. - PubMed
-
- Brooks GA. Lactate: link between glycolytic and oxidative metabolism. Sports Med. 2007;37:341–343. - PubMed
-
- Bergersen LH. Is lactate food for neurons? Comparison of monocarboxylate transporter subtypes in brain and muscle. Neuroscience. 2007;145:11–19. - PubMed
-
- Brooks GA. Lactate shuttles in nature. Biochem Soc Trans. 2002;30:258–264. - PubMed
-
- Brooks GA. Current concepts in lactate exchange. Med Sci Sports Exerc. 1991;23:895–906. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 CA075503/CA/NCI NIH HHS/United States
- R01 CA098779/CA/NCI NIH HHS/United States
- R01-CA-120876/CA/NCI NIH HHS/United States
- R01 CA120876/CA/NCI NIH HHS/United States
- R01-CA-70896/CA/NCI NIH HHS/United States
- R01-CA-098779/CA/NCI NIH HHS/United States
- R01-CA-86072/CA/NCI NIH HHS/United States
- R01-AR-055660/AR/NIAMS NIH HHS/United States
- R01-CA-080250/CA/NCI NIH HHS/United States
- R01 CA070896/CA/NCI NIH HHS/United States
- R01 EY012042/EY/NEI NIH HHS/United States
- R01 CA107382/CA/NCI NIH HHS/United States
- P30 CA056036/CA/NCI NIH HHS/United States
- P30-CA-56036/CA/NCI NIH HHS/United States
- R01-CA-107382/CA/NCI NIH HHS/United States
- R01 AR055660/AR/NIAMS NIH HHS/United States
- R01-CA-75503/CA/NCI NIH HHS/United States
- R01 CA080250/CA/NCI NIH HHS/United States
- R01 CA086072/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
