Passive immunization reduces behavioral and neuropathological deficits in an alpha-synuclein transgenic model of Lewy body disease
- PMID: 21559417
- PMCID: PMC3084838
- DOI: 10.1371/journal.pone.0019338
Passive immunization reduces behavioral and neuropathological deficits in an alpha-synuclein transgenic model of Lewy body disease
Retraction in
-
Retraction: Passive Immunization Reduces Behavioral and Neuropathological Deficits in an Alpha-Synuclein Transgenic Model of Lewy Body Disease.PLoS One. 2024 Nov 13;19(11):e0313936. doi: 10.1371/journal.pone.0313936. eCollection 2024. PLoS One. 2024. PMID: 39536007 Free PMC article. No abstract available.
Abstract
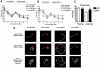
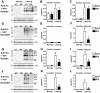
Dementia with Lewy bodies (DLB) and Parkinson's Disease (PD) are common causes of motor and cognitive deficits and are associated with the abnormal accumulation of alpha-synuclein (α-syn). This study investigated whether passive immunization with a novel monoclonal α-syn antibody (9E4) against the C-terminus (CT) of α-syn was able to cross into the CNS and ameliorate the deficits associated with α-syn accumulation. In this study we demonstrate that 9E4 was effective at reducing behavioral deficits in the water maze, moreover, immunization with 9E4 reduced the accumulation of calpain-cleaved α-syn in axons and synapses and the associated neurodegenerative deficits. In vivo studies demonstrated that 9E4 traffics into the CNS, binds to cells that display α-syn accumulation and promotes α-syn clearance via the lysosomal pathway. These results suggest that passive immunization with monoclonal antibodies against the CT of α-syn may be of therapeutic relevance in patients with PD and DLB.
Conflict of interest statement
Figures
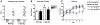
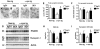

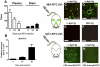
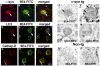
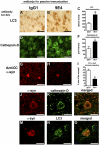

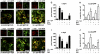
References
-
- McKeith IG. Spectrum of Parkinson's disease, Parkinson's dementia, and Lewy body dementia. Neurol Clin. 2000;18:865–902. - PubMed
-
- Weinreb P, Zhen W, Poon A, Conway K, Lansbury PJ. NACP, a protein implicated in Alzheimer's disease and learning, is natively unfolded. Biochem. 1996;35:13709–13715. - PubMed
-
- Iwai A, Masliah E, Yoshimoto M, Ge N, Flanagan L, et al. The precursor protein of non-A beta component of Alzheimer's disease amyloid is a presynaptic protein of the central nervous system. Neuron. 1995;14:467–475. - PubMed
-
- Hashimoto M, Hsu LJ, Xia Y, Takeda A, Sisk A, et al. Oxidative stress induces amyloid-like aggregate formation of NACP/alpha-synuclein in vitro. Neuroreport. 1999;10:717–721. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

