The polymodal ion channel transient receptor potential vanilloid 4 modulates calcium flux, spiking rate, and apoptosis of mouse retinal ganglion cells
- PMID: 21562271
- PMCID: PMC3109951
- DOI: 10.1523/JNEUROSCI.0359-11.2011
The polymodal ion channel transient receptor potential vanilloid 4 modulates calcium flux, spiking rate, and apoptosis of mouse retinal ganglion cells
Abstract
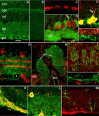
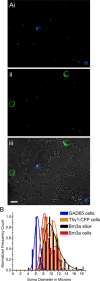
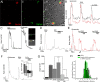
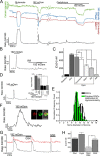
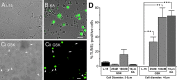
Sustained increase in intraocular pressure represents a major risk factor for eye disease, yet the cellular mechanisms of pressure transduction in the posterior eye are essentially unknown. Here we show that the mouse retina expresses mRNA and protein for the polymodal transient receptor potential vanilloid 4 (TRPV4) cation channel known to mediate osmotransduction and mechanotransduction. TRPV4 antibodies labeled perikarya, axons, and dendrites of retinal ganglion cells (RGCs) and intensely immunostained the optic nerve head. Müller glial cells, but not retinal astrocytes or microglia, also expressed TRPV4 immunoreactivity. The selective TRPV4 agonists 4α-PDD and GSK1016790A elevated [Ca2+]i in dissociated RGCs in a dose-dependent manner, whereas the TRPV1 agonist capsaicin had no effect on [Ca2+](RGC). Exposure to hypotonic stimulation evoked robust increases in [Ca2+](RGC). RGC responses to TRPV4-selective agonists and hypotonic stimulation were absent in Ca2+ -free saline and were antagonized by the nonselective TRP channel antagonists Ruthenium Red and gadolinium, but were unaffected by the TRPV1 antagonist capsazepine. TRPV4-selective agonists increased the spiking frequency recorded from intact retinas recorded with multielectrode arrays. Sustained exposure to TRPV4 agonists evoked dose-dependent apoptosis of RGCs. Our results demonstrate functional TRPV4 expression in RGCs and suggest that its activation mediates response to membrane stretch leading to elevated [Ca2+]i and augmented excitability. Excessive Ca2+ influx through TRPV4 predisposes RGCs to activation of Ca2+ -dependent proapoptotic signaling pathways, indicating that TRPV4 is a component of the response mechanism to pathological elevations of intraocular pressure.
Figures
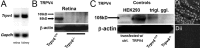


References
-
- Agar A, Li S, Agarwal N, Coroneo MT, Hill MA. Retinal ganglion cell line apoptosis induced by hydrostatic pressure. Brain Res. 2006;1086:191–200. - PubMed
-
- Becker D, Blase C, Bereiter-Hahn J, Jendrach M. TRPV4 exhibits a functional role in cell-volume regulation. J Cell Sci. 2005;118:2435–2440. - PubMed
-
- Benfenati V, Amiry-Moghaddam M, Caprini M, Mylonakou MN, Rapisarda C, Ottersen OP, Ferroni S. Expression and functional characterization of transient receptor potential vanilloid-related channel 4 (TRPV4) in rat cortical astrocytes. Neuroscience. 2007;148:876–892. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
