Regression of recurrent malignant gliomas with convection-enhanced delivery of topotecan
- PMID: 21562434
- PMCID: PMC4940854
- DOI: 10.1227/NEU.0b013e3182233e24
Regression of recurrent malignant gliomas with convection-enhanced delivery of topotecan
Abstract
Background: Convection-enhanced delivery of chemotherapeutics for the treatment of malignant glioma is a technique that delivers drugs directly into a tumor and the surrounding interstitium through continuous, low-grade positive-pressure infusion. This allows high local concentrations of drug while overcoming the limitations imposed by toxicity and the blood-brain barrier in systemic therapies that prevent the use of many potentially effective drugs.
Objective: To examine the safety profile of a conventional chemotherapeutic agent, topotecan, via convection-enhanced delivery in the treatment of recurrent malignant gliomas and secondarily to assess radiographic response and survival.
Methods: We performed a prospective, dose-escalation phase Ib study of the topoisomerase-I inhibitor topotecan given by convection-enhanced delivery in patients with recurrent malignant gliomas.
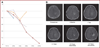
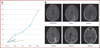
Results: Significant antitumor activity as described by radiographic changes and prolonged overall survival with minimal drug-associated toxicity was demonstrated. A maximum tolerated dose was established for future phase II studies.
Conclusion: Topotecan by convection-enhanced delivery has significant antitumor activity at concentrations that are nontoxic to normal brain. The potential for use of this therapy as a generally effective treatment option for malignant gliomas will be tested in subsequent phase II and III trials.
Figures

References
-
- Barker FG, II, Chang SM, Gutin PH, et al. Survival and functional status after resection of recurrent glioblastoma multiforme. Neurosurgery. 1998;42(4):709–720. - PubMed
-
- Broaddus WC, Prabhu SS, Gillies GT, et al. Distribution and stability of antisense phosphorothioate oligonucleotides in rodent brain following direct intraparenchymal controlled-rate infusion. J Neurosurg. 1998;88(4):734–742. - PubMed
-
- Groothuis DR, Benalcazar H, Allen CV, et al. Comparison of cytosine arabinoside delivery to rat brain by intravenous, intrathecal, intraventricular and intraparenchymal routes of administration. Brain Res. 2000;856(1–2):281–290. - PubMed
-
- Kaiser MG, Parsa AT, Fine RL, Hall JS, Chakrabarti I, Bruce JN. Tissue distribution and antitumor activity of topotecan delivered by intracerebral clysis in a rat glioma model. Neurosurgery. 2000;47(6):1391–1398. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

