Perivascular adipose tissue from human systemic and coronary vessels: the emergence of a new pharmacotherapeutic target
- PMID: 21564083
- PMCID: PMC3315039
- DOI: 10.1111/j.1476-5381.2011.01479.x
Perivascular adipose tissue from human systemic and coronary vessels: the emergence of a new pharmacotherapeutic target
Abstract
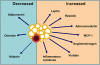
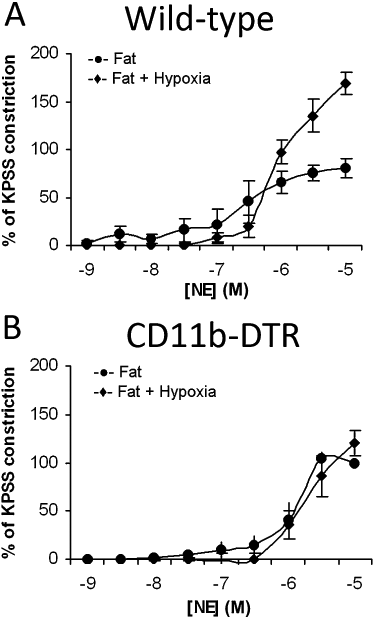
Fat cells or adipocytes are distributed ubiquitously throughout the body and are often regarded purely as energy stores. However, recently it has become clear that these adipocytes are engine rooms producing large numbers of metabolically active substances with both endocrine and paracrine actions. White adipocytes surround almost every blood vessel in the human body and are collectively termed perivascular adipose tissue (PVAT). It is now well recognized that PVAT not only provides mechanical support for any blood vessels it invests, but also secretes vasoactive and metabolically essential cytokines known as adipokines, which regulate vascular function. The emergence of obesity as a major challenge to our healthcare systems has contributed to the growing interest in adipocyte dysfunction with a view to discovering new pharmacotherapeutic agents to help rescue compromised PVAT function. Very few PVAT studies have been carried out on human tissue. This review will discuss these and the hypotheses generated from such research, as well as highlight the most significant and clinically relevant animal studies showing the most pharmacological promise.
Linked articles: This article is part of a themed section on Fat and Vascular Responsiveness. To view the other articles in this section visit http://dx.doi.org/10.1111/bph.2012.165.issue-3.
© 2011 The Authors. British Journal of Pharmacology © 2011 The British Pharmacological Society.
Figures


References
-
- Asayama K, Hayashibe H, Dobashi K, Uchida N, Nakane T, Kodera K, et al. Decrease in serum adiponectin level due to obesity and visceral fat accumulation in children. Obesity. 2003;11:1072–1079. - PubMed
-
- Bakker EN, Pistea A, Spaan JA, Rolf T, de Vries CJ, van Rooijen N, et al. Flow-dependent remodeling of small arteries in mice deficient for tissue-type transglutaminase: possible compensation by macrophage-derived factor XIII. Circ Res. 2006;99:86–92. - PubMed
-
- Belin de Chantemele EJ, Vessieres E, Guihot AL, Toutain B, Maquignau M, Loufrani L, et al. Type 2 diabetes severely impairs structural and functional adaptation of rat resistance arteries to chronic changes in blood flow. Cardiovasc Res. 2009;81:788–796. - PubMed
-
- Berg AH, Combs TP, Du X, Brownlee M, Scherer PE. The adipocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat Med. 2001;7:947–953. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

