E2F-1- and E2Ftr-mediated apoptosis: the role of DREAM and HRK
- PMID: 21564512
- PMCID: PMC3822935
- DOI: 10.1111/j.1582-4934.2011.01338.x
E2F-1- and E2Ftr-mediated apoptosis: the role of DREAM and HRK
Abstract
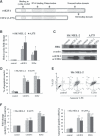
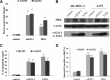
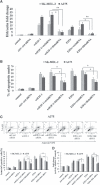
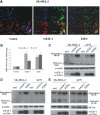
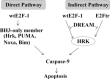
E2F-1-deleted mutant, 'truncated E2F' (E2Ftr, E2F-1[1-375]), lacking the carboxy-terminal transactivation domain, was shown to be more potent at inducing cancer cell apoptosis than wild-type E2F-1 (wtE2F-1; full-length E2F-1). Mechanisms by which wtE2F-1 and E2Ftr induce apoptosis, however, are not fully elucidated. Our study demonstrates molecular effects of pro-apoptotic BH3-only Bcl-2 family member Harakiri (Hrk) in wtE2F-1- and E2Ftr-induced melanoma cell apoptosis. We found that Hrk mRNA and Harakiri (HRK) protein expression was highly up-regulated in melanoma cells in response to wtE2F-1 and E2Ftr overexpression. HRK up-regulation did not require the E2F-1 transactivation domain. In addition, Hrk gene up-regulation and HRK protein expression did not require p53 in cancer cells. Hrk knockdown by Hrk siRNA was associated with significantly reduced wtE2F-1- and E2Ftr-induced apoptosis. We also found that an upstream factor, 'downstream regulatory element antagonist modulator' (DREAM), may be involved in HRK-mediated apoptosis in response to wtE2F-1 and E2Ftr overexpression. DREAM expression levels increased following wtE2F-1 and E2Ftr overexpression. Western blotting detected increased DREAM primarily in dimeric form. The homodimerization of DREAM resulting from wtE2F-1 and E2Ftr overexpression may contribute to the decreased binding activity of DREAM to the 3'-untranslated region of the Hrk gene as shown by electromobility shift assay. Results showed wtE2F-1- and E2Ftr-induced apoptosis is partially mediated by HRK. HRK function is regulated in response to DREAM. Our findings contribute to understanding the mechanisms that regulate wtE2F-1- and E2Ftr-induced apoptosis and provide insights into the further evaluation of how E2Ftr-induced apoptosis may be used for therapeutic gain.
© 2012 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures


References
-
- Phillips AC, Vousden KH. E2F-1 induced apoptosis. Apoptosis. 2001;6:173–82. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

