Doppler velocity measurements from large and small arteries of mice
- PMID: 21572013
- PMCID: PMC3154663
- DOI: 10.1152/ajpheart.00320.2011
Doppler velocity measurements from large and small arteries of mice
Abstract
With the growth of genetic engineering, mice have become increasingly common as models of human diseases, and this has stimulated the development of techniques to assess the murine cardiovascular system. Our group has developed nonimaging and dedicated Doppler techniques for measuring blood velocity in the large and small peripheral arteries of anesthetized mice. We translated technology originally designed for human vessels for use in smaller mouse vessels at higher heart rates by using higher ultrasonic frequencies, smaller transducers, and higher-speed signal processing. With these methods one can measure cardiac filling and ejection velocities, velocity pulse arrival times for determining pulse wave velocity, peripheral blood velocity and vessel wall motion waveforms, jet velocities for the calculation of the pressure drop across stenoses, and left main coronary velocity for the estimation of coronary flow reserve. These noninvasive methods are convenient and easy to apply, but care must be taken in interpreting measurements due to Doppler sample volume size and angle of incidence. Doppler methods have been used to characterize and evaluate numerous cardiovascular phenotypes in mice and have been particularly useful in evaluating the cardiac and vascular remodeling that occur following transverse aortic constriction. Although duplex ultrasonic echo-Doppler instruments are being applied to mice, dedicated Doppler systems are more suitable for some applications. The magnitudes and waveforms of blood velocities from both cardiac and peripheral sites are similar in mice and humans, such that much of what is learned using Doppler technology in mice may be translated back to humans.
Figures


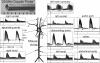

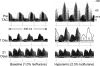
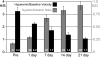

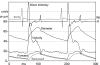
Similar articles
-
Noninvasive cardiovascular phenotyping in mice.ILAR J. 2002;43(3):147-58. doi: 10.1093/ilar.43.3.147. ILAR J. 2002. PMID: 12105382 Review.
-
Feasibility of dual Doppler velocity measurements to estimate volume pulsations of an arterial segment.Ultrasound Med Biol. 2010 Jul;36(7):1169-75. doi: 10.1016/j.ultrasmedbio.2010.04.002. Ultrasound Med Biol. 2010. PMID: 20620703 Free PMC article.
-
In vivo hemodynamic evaluation based on transverse Doppler measurements of blood velocities and vessel diameter.IEEE Trans Biomed Eng. 1999 Sep;46(9):1074-80. doi: 10.1109/10.784138. IEEE Trans Biomed Eng. 1999. PMID: 10493070
-
Doppler evaluation of peripheral vascular adaptations to transverse aortic banding in mice.Ultrasound Med Biol. 2003 Sep;29(9):1281-9. doi: 10.1016/s0301-5629(03)00986-4. Ultrasound Med Biol. 2003. PMID: 14553805
-
Blood flow: insights from ultrasound.Proc Inst Mech Eng H. 1990;204(1):1-20. doi: 10.1243/PIME_PROC_1990_204_223_02. Proc Inst Mech Eng H. 1990. PMID: 2191674 Review.
Cited by
-
Mouse Cardiovascular Imaging.Curr Protoc. 2024 Sep;4(9):e1116. doi: 10.1002/cpz1.1116. Curr Protoc. 2024. PMID: 39222027 Review.
-
Ultrasound detection of altered placental vascular morphology based on hemodynamic pulse wave reflection.Am J Physiol Heart Circ Physiol. 2017 May 1;312(5):H1021-H1029. doi: 10.1152/ajpheart.00791.2016. Epub 2017 Mar 31. Am J Physiol Heart Circ Physiol. 2017. PMID: 28364018 Free PMC article.
-
Resting blood flow in the skin: does it exist, and what is the influence of temperature, aging, and diabetes?J Diabetes Sci Technol. 2012 May 1;6(3):674-85. doi: 10.1177/193229681200600324. J Diabetes Sci Technol. 2012. PMID: 22768900 Free PMC article. Review.
-
Vascular and parenchymal amyloid pathology in an Alzheimer disease knock-in mouse model: interplay with cerebral blood flow.Mol Neurodegener. 2014 Aug 9;9:28. doi: 10.1186/1750-1326-9-28. Mol Neurodegener. 2014. PMID: 25108425 Free PMC article.
-
In Vivo Flow Cytometry of Extremely Rare Circulating Cells.Sci Rep. 2019 Mar 4;9(1):3366. doi: 10.1038/s41598-019-40143-2. Sci Rep. 2019. PMID: 30833671 Free PMC article.
References
-
- Aristizabal O, Christopher DA, Foster FS, Turnbull DH. 40-MHz echocardiographic scanner for cardiovascular assessment of mouse embryos. Ultrasound Med Biol 24: 1407–1417, 1998 - PubMed
-
- Asmar R, Benetos A, Topouchian J, Laurent P, Pannier B, Brisac AM, Target R, Levy BI. Assessment of arterial distensibility by automatic pulse wave velocity measurement. Validation and clinical studies. Hypertension 26: 485–490, 1995 - PubMed
-
- Baker DW. Pulsed ultrasonic Doppler blood flow sensing. IEEE Trans Sonics Ultrason SU-17: 170–185, 1970
-
- Bonnin P, Sabaa N, Flamant M, Debbabi H, Tharaux PL. Ultrasound imaging of renal vaso-occlusive events in transgenic sickle mice exposed to hypoxic stress. Ultrasound Med Biol 34: 1076–1084, 2008 - PubMed
-
- Bonnin P, Villemain A, Vincent F, Debbabi H, Silvestre JS, Contreses JO, Levy BI, Tobelem G, Dupuy E. Ultrasonic assessement of hepatic blood flow as a marker of mouse hepatocarcinoma. Ultrasound Med Biol 33: 561–570, 2007 - PubMed
Publication types
MeSH terms
Grants and funding
- HL-89792/HL/NHLBI NIH HHS/United States
- HL-57068/HL/NHLBI NIH HHS/United States
- HL-42267/HL/NHLBI NIH HHS/United States
- AG-15568/AG/NIA NIH HHS/United States
- R01 HL022512/HL/NHLBI NIH HHS/United States
- HL-42313/HL/NHLBI NIH HHS/United States
- P41 RR011795/RR/NCRR NIH HHS/United States
- HL-52364/HL/NHLBI NIH HHS/United States
- P41 EB002182/EB/NIBIB NIH HHS/United States
- R01 HL089792/HL/NHLBI NIH HHS/United States
- HL-13870/HL/NHLBI NIH HHS/United States
- HL-42550/HL/NHLBI NIH HHS/United States
- AG-13251/AG/NIA NIH HHS/United States
- HL-22512/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources

