In vivo ultrastructural analysis of the intimate relationship between polymorphonuclear leukocytes and the chlamydial developmental cycle
- PMID: 21576327
- PMCID: PMC3147583
- DOI: 10.1128/IAI.00200-11
In vivo ultrastructural analysis of the intimate relationship between polymorphonuclear leukocytes and the chlamydial developmental cycle
Abstract
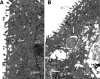
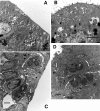
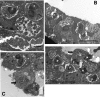
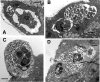
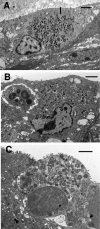
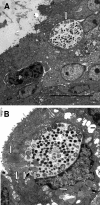
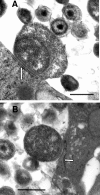
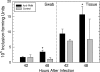
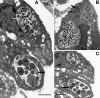
We utilized a recently developed model of intracervical infection with Chlamydia muridarum in the mouse to elicit a relatively synchronous infection during the initial developmental cycle in order to examine at the ultrastructural level the development of both the chlamydial inclusion and the onset of the inflammatory response. At 18 h after infection, only a few elementary bodies attached to cells were visible, as were an occasional intracellular intermediate body and reticulate body. By 24 h, inclusions had 2 to 5 reticulate bodies and were beginning to fuse. A few polymorphonuclear leukocytes (PMNs) were already present in the epithelium in the vicinity of and directly adjacent to infected cells. By 30 h, the inclusions were larger and consisted solely of reticulate bodies, but by 36 to 42 h, they contained intermediate bodies and elementary bodies as well. Many PMNs were adjacent to or actually inside infected cells. Chlamydiae appeared to exit the cell either (i) through disintegration of the inclusion membrane and rupture of the cell, (ii) by dislodgement of the cell from the epithelium by PMNs, or (iii) by direct invasion of the infected cell by the PMNs. When PMNs were depleted, the number of released elementary bodies was significantly greater as determined both visually and by culture. Interestingly, depletion of PMNs revealed the presence of inclusions containing aberrant reticulate bodies, reminiscent of effects seen in vitro when chlamydiae are incubated with gamma interferon. In vivo evidence for the contact-dependent development hypothesis, a potential mechanism for triggering the conversion of reticulate bodies to elementary bodies, and for translocation of lipid droplets into the inclusion is also presented.
Figures
References
-
- Bedson S. P., Bland J. O. W. 1932. A morphological study of psittacosis virus, with the description of a developmental cycle. Brit. J. Exp. Pathol. 13:461–466
-
- Byrne G. I., Ojcius D. M. 2004. Chlamydia and apoptosis: life and death decisions of an intracellular pathogen. Nat. Rev. Microbiol. 2:802–808 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

